Narmadha Kuppuswami, MD, FACOG1, Suresh Subramanian, MS, PhD2, Karenna J. Groff3, Radha Rani Ravichandran, BDS4
Affiliations: 1Advocate Good Samaritan Hospital, Downers Grove, IL, USA; 2Harvard T. H. Chan School of Public Health, Boston, MA, USA; 3Massachusetts Institute of Technology (MIT), Department of Biological Engineering, Cambridge, MA, USA; 4WONDER Clinical Coordinator, Erode, India
Corresponding Author: Narmadha Kuppuswami, Email: narmadhakupp@gmail.com
Keywords: Eclampsia, Maternal Health, Maternal Mortality, MEOWS, Telehealth
Section: Proof of Concept
Introduction: In this article, we describe a pilot telehealth project for identifying women at risk of developing serious complications early and for instituting timely, appropriate, and up-to-date management even in situations with limited resources and skilled obstetric services. Maternal mortality remains unacceptably high, with less than two-thirds of the signatories to the 2015 Millennium Development Goals achieving the outlined 75% reduction in maternal mortality ratio (MMR) from 1990 to 2015. Looking forward to 2030, the Sustainable Development Goals (SDGs) lay out a target of reducing the MMR in every country to below 70 per 100,000 live births. This will require progress in low-and-middle-income countries at a rate much greater than that seen over the past 15 years. Given that 94% of the global maternal deaths occur in low- and-middle-income countries, a solution to meet the unique challenges of these countries will be necessary to achieve the SDG. The Women’s Obstetrical Neonatal Death and Reduction (WONDER) telehealth system described here offers a potential telehealth solution to reduce mortality and morbidity rates in resource-limited environments by early identification of risk indicators and initiation of care.
Materials and methods: The WONDER system consists of a cloud-based electronic health record with a Clinical Decision Support tool and a color-coded alert system. The Clinical Decision Support tool is based upon Maternal Early Warning Signs and provides real-time assistance to caregivers via relevant national treatment guidelines. This system uses inexpensive computing hardware, displays, and cell-phone technology. This system was tested in a 2-year pilot study in India. A total of 15,184 patients were monitored during labor and the postpartum period.
Results: Within limitations of the study, the incidence of in-hospital eclampsia was reduced by 91.7%, and in 95% of cases, timely treatment was started within an hour of identifying the abnormality in vital signs. Maternal mortality was reduced by 50.1% over local benchmark figures.
Conclusions: The WONDER system identified at-risk patients, directed skilled care to those patients at risk for complications, and helped to institute effective, timely treatment, demonstrating a potential solution for women in resource-limited locations.
This pilot study describes a novel telehealth solution for the improved treatment of pregnant women in resource-poor settings. The system continuously identifies women at risk of developing serious complication in labor or postpartum and helps institute timely, appropriate care even in the absence of physicians.
Maternal mortalities and morbidities disproportionately affect women in low- and middle-income countries (LMICs), with 94% of all maternal deaths reported in low-resource areas.1 Reported as deaths per 100,000 live births, the number of maternal mortalities in 2017 ranged from a low of 2 in a handful of European countries to a high of over 1,100 in South Sudan. Overall, the risk of maternal death is over 100 times greater for women in sub-Saharan Africa than for those in Europe.2 At the Millennium Summit in September 2000, 189 countries affirmed their commitment to the Millennium Development Goals (MDGs), with the aim of tackling problems that harm the world’s poorest and most vulnerable populations. MDG 5 focused on maternal mortality and called for a 75% reduction in the maternal mortality ratio (MMR) between 1990 and 2015, a figure that was met by less than two-thirds of the signatories.3 In fact, as of 2017, a third of the countries in the world had not reached their MDG for reduction in MMR. In 2015 and as a follow up to the MDGs, 193 countries came together to establish and ratify 17 Sustainable Development Goals (SDGs) with a target date of 2030.4 Goal number 3 of the SDG covers maternal health and specifically lays out the objective of reducing worldwide MMR to 70 or below by 2030.4 Achieving this goal requires a sustained, annual reduction in maternal mortality of 7.5% from 2015 to 2030, a number that is over twice the improvement rates that were achieved during the 2000–2015 period.
Worldwide, maternal mortality and morbidity rates are found to be high in countries with a shortage of healthcare infrastructure and skilled providers, where access to health care is limited. The high prevalence of maternal mortality is now, for the most part, concentrated in sub-Saharan Africa and South Asia.4,5 The countries of sub-Saharan Africa collectively account for 66% of all maternal deaths worldwide, while in South Asia, India alone accounts for 16% of the global tally.5 This article outlines a low-cost telehealth approach and the results of a 2-year pilot project implemented in the state of Tamil Nadu (TN), India to target maternal mortality. The project outlined here has applicability to India, other parts of South Asia, and sub-Saharan Africa.
India has demonstrated an impressive track record of reducing maternal mortality from approximately 556 in 1990 to 130 in 2016.6 This improvement has come about through a range of multi-sectoral government interventions targeting improvements in the three major causal drivers of maternal mortality. These included the following: institutional births increased from 38.7% in 2000 to 78.9% in 2015, the proportion of girls getting married below the age of 18 reduced from 47% to 27% by 2015, and female literacy improved from 54% in 2000 to 63% in 2015.6
This progress, however, may not be reflective of the situation for women living in the poorest areas of India. For instance, in Uttar Pradesh, a state in the northern part of India, MMR varies from 280 to 1,180 deaths per 100,000 live births, with the highest MMR being present in the rural districts.7,8 It can be further argued that the progress achieved, to date, represents the most readily accessible opportunities to reducing MMR and the difficult task of reducing maternal mortality rates in rural and less economically developed regions now lies ahead for India.6 This only increases the urgency for public health solutions targeting maternal health, which are specifically designed for rural and resource-poor areas.
In examining antecedents for maternal mortality, multiple studies reveal delay in triage, transport, and treatment as potential factors in the increase of maternal deaths.9–11 One of the major factors contributing to maternal deaths regardless of resources is the delay in instituting appropriate skilled obstetric care. Because of this delay in care, even preventable problems escalate quickly. For instance, studies have highlighted that preeclampsia often goes unrecognized until it manifests as a crisis during labor, making eclampsia a leading cause of maternal death.12,13
Eclampsia is defined as the onset of generalized, tonic-clonic seizures and/or unexplained coma during pregnancy or postpartum in a woman with signs or symptoms of severe preeclampsia and without any underlying seizure disorders. This multisystem disorder affects almost every organ in the body and contributes to significant maternal and neonatal mortality and morbidity.14 Approximately 50,000 women die of eclampsia each year globally, with close to 99% reported in LMICs.12 The risk of a woman in a developing country dying of preeclampsia or eclampsia is over 300 times that of a woman in a developed country.15 In a systematic review covering 39 million women from 40 countries, Abalos et al. estimated the incidence of preeclampsia to be 4.6% (95% uncertainty range 2.7–8.2) and eclampsia to be 1.4% (95% uncertainty range 1.0–2.0) with a wide variation across regions.16 In Africa and Asia, hypertensive diseases accounted for 9% of maternal deaths, whereas in Latin America and the Caribbean, the figure is over 25%.17 In India, several reports indicate that the incidence of eclampsia has remained steady over a period of 40–50 years and ranges from 0.179% to 5%, with the average being 1.5%.18 In 2019, Vousden et al., in their analysis of a cluster of randomized controlled trials of hypertensive disorders of pregnancy across 10 low- and middle-income geographic regions in eight countries, reported that 6.9% of women with eclampsia died, and highlighted the inequality and inequity that persist in health care for women with hypertensive disorders of pregnancy.19 Various authors suggest that interventions for recognizing at-risk patients and processes to initiate early referral to specialists are critical to reduce deaths from preeclampsia or eclampsia.15 Telehealth solutions offer an important opportunity to deploy rapid and cost-effective interventions to address these issues in LMICs.
Women who die during childbirth do not go directly from a healthy state to the verge of death. Rather, there is a crucial transition period during which healthcare providers can intervene and institute appropriate treatment to prevent the situation from deteriorating. This is more readily possible in a clinical setting with full resources; however, it is difficult where resources are limited as is often the case in level I hospitals or in Primary Health care Centers (PHCs) with no skilled services. Identifying this crucial time period through monitoring health indicators in real or near-real time and instituting interventions before the patient’s condition worsens are critical to saving maternal lives. By applying telehealth solutions to this problem, that critical period can be identified even in the absence of a skilled physician, making care more equitable and accessible to women in rural, low-income areas.
In 2014, the Women’s Obstetrical Neonatal Death Evaluation and Reduction (WONDER) Telehealth Solution was founded, a US-based company with the goal of developing telemedicine and health solutions to address the high rates of maternal mortality and morbidity around the world. The focus of this proof-of-concept study was TN, the state with the second lowest MMR rankings in India. TN offered middle-level resources and infrastructure, and was typical of states in India. The objective of this study was to implement the telehealth approach in TN before migrating the solution to other states in India and then to sub-Saharan Africa. At the time of this writing, the solution has been migrated to the state of Bihar where over 60,000 pregnant women are enrolled in a large-scale pilot. This study, however, will focus on the pilot project in TN, the results generated, and the lessons learned. The WONDER project team included partners with extensive experience in high-risk obstetrics, electronic health records (EHRs), telemedicine, hardware and software design, and technical service and support capability in rural India.
The WONDER Telehealth Solution tested in TN included three components: (1) a smart EHR with clinical alerts and treatment guidelines; (2) a WONDER App, a mobile-based clinical Decision Support tool that at its core algorithmically scored patients in real time; and (3) a telecommunications capability using an existing and inexpensive infrastructure between remote PHCs and the District Hospital. The system was piloted, with three General Hospitals (GHs) and five surrounding PHCs extending out to a distance of 70 km. The electronic health-record system was developed based on the American College of Obstetricians and Gynecologists (ACOG), National Health Service (NHS), and World Health Organization (WHO) standards and was current on The International Classification of Diseases (ICD) 10 classifications. The system fully complied with all The Centers for Medicare and Medicaid Services (CMS) and The Health Insurance Portability and Accountability Act of 1996 (HIPAA) requirements. In keeping with the objective of speed, security, and low-cost, all generated data were stored securely in the cloud. Utilizing a cloud-based platform also permitted multiple doctors at different sites, including those from remote locations, to use the system simultaneously and allowed users to quickly and efficiently manage patients’ personal information. The system also used the readily available mobile technology with two-way communications (the WONDER platform and capabilities have been expanded beyond what was tested in TN by including “Internet of Things” (IOT) capabilities to integrate monitoring devices into the EHR and automatically upload values. However, for the purposes of this report, these details will not be included, except in the Discussion section).
The core of WONDER is an algorithm based on the Maternal Early Obstetric Warning Signs (MEOWS), a scoring and presentation system highly recommended by the ACOG Council on Patient safety in Women’s Health Care and Royal College of Obstetricians and Gynecologists of UK (Table 1). The criteria were modified to address local issues and availability.
Table 1. Maternal health scoring criteria outlined by the Maternal Early Obstetric Warning Score System (MEOWS) modified to address local needs and requirements
| Clinical parameter | Yellow alert | Red alert |
|---|---|---|
| Systolic blood pressure (mm Hg) | 90–99 or 130–150 | <90 or >150 |
| Diastolic blood pressure (mm Hg) | 50–59 or 90–99 | <50 or >100 |
| Heart rate (beats per minute) | 40–50 or 100–120 | <40 or >120 |
| Respiratory rate (breaths per minute) | 21–25 | <10 or >26 |
| Oxygen saturation (%) | <95% | |
| Temperature | 35–36°C 100–101°F |
<35°C or >37.5°C >101°F |
| Urine output | <33 CC/hour | |
| Fetal heart rate | <110 | >160 |
| Hemoglobin (grams) | 8–9 | <8 |
| HIV status | Positive | |
| Hepatitis HBsAg | Positive |
HBsAg, hepatitis B surface antigen; HIV, human immunodeficiency virus.
The WONDER algorithm works on the core concept that in many instances, individual vital signs (VS) of a woman in labor or postpartum may not indicate the presence or possibility of a crisis but when combined with patient history and readings on other vitals, they identify and forecast a potential emergency with a high level of accuracy. The WONDER EHR and APP system sets three color codes: green for normal vitals, yellow for a patient or situation that needs attention, and red for a crisis situation requiring immediate attention. An audible alarm sounds whenever a patient is scored yellow or red. For scoring each patient and providing the appropriate color codes and any necessary alarms, the WONDER system inputs five VS: blood pressure, pulse rate, temperature, respiratory rate, and oxygen saturation.
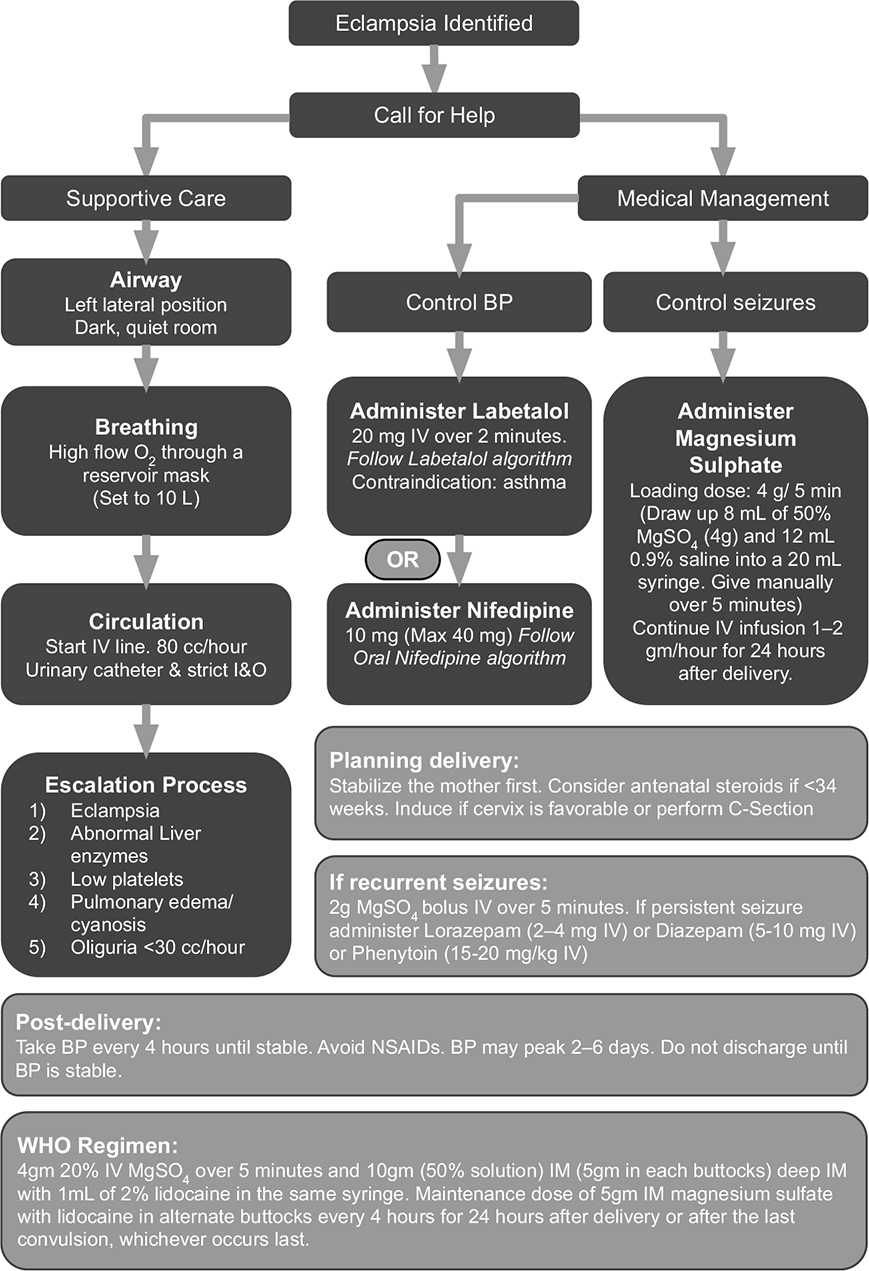
In addition, to ensure that necessary information is ready at hand for physicians and nurses to attend to alarms, WONDER includes a full decision support system. The decision support capability works with the patient’s history and current VS to serve up a possible diagnosis and up-to-date treatment guidelines, including, medications, dosage, administration protocol, side effects, and contraindications, with links to the actual recommendations from various organizations. These guidelines have been built and updated based on WHO, ACOG, and National Institute for Health and Care Excellence guidelines. This decision support system is designed to address the three major causes that collectively account for 52% of all maternal deaths: preeclampsia or eclampsia, postpartum hemorrhage, and sepsis.20 For preeclampsia or eclampsia and postpartum hemorrhage, the program incorporates the appropriate guidelines from the ACOG Council on Patient safety and California Maternal Quality Care Collaborative. For sepsis, the international guidelines from the “Surviving Sepsis” campaign were used. In addition, the Government of India requires doctors to follow the Federation of Obstetric and Gynecological Society of India (FOGSI) guidelines for the treatment of preeclampsia and eclampsia. The WONDER system is flexible to add any protocols set forth by any given institution or governing body and provides physicians links to the references and the option to apply whichever protocol they are required to use or prefer. The decision support system within WONDER was amended for this pilot project to align completely with all of the above guidelines, and an example of the guidelines provided by WONDER to providers is shown in Figure 1. While WONDER was designed to address preeclampsia or eclampsia, postpartum hemorrhage, and sepsis, anticipating difficulties in getting access to physicians to participate in a 2-year project, and keeping in mind limitations on funds and personnel, we decided to focus this project exclusively on preventing in-hospital eclampsia.
Figure 1—WONDER Eclampsia Algorithm modified from NHS, UK, and World Health Organization (WHO) guidelines.
At the satellite PHCs with limited facilities and skilled care, the system provided guidelines, order sets, and instructions for management of acute obstetric emergencies to the nurses or auxiliary nurse midwives when a skilled provider was not available. In addition, as patient records were stored in the cloud, medical records of patients in remote PHCs were available in the GH as soon as they were created. In situations involving an “unstable” patient in a remote PHC, the system provided the nurse or physician at the GH an alert to instruct the care provider in the PHC with instructions on how to stabilize the patient prior to transport. Once the patient was in transit, a flashing ambulance sign and alarm on the dashboard at the GH alerted the physicians and staff in the GH to make necessary preparations to take care of the incoming acute obstetric patient.
The objective of this pilot project was to (1) examine the viability of the WONDER telehealth system to identify patients at risk of complications during labor early and initiate rapid medical intervention, and (2) test the telehealth system in a live low resource setting to identify opportunities for modification and improvement. The 2-year pilot study ran from July 1, 2017 to June 30, 2019. The WONDER telehealth system was implemented in April 1, 2017; the system was turned on for live patient monitoring and data collection on July 1st after a period of training, minor software adjustments and hardware stabilization.
The WONDER system was implemented as an enhancement to the existing maternal healthcare capability and infrastructure and not as a substitute. No government medical resources were redirected from their assigned tasks to manage the WONDER implementation, and all resources in the WONDER project were positioned outside of the government resources. Prior to commencing implementation of the project, WONDER administrative staff received permission from the top administrative official of the Erode district in TN, India, who, in turn, secured clearance from the National Health Mission Director of the State to permit the study to proceed. As mentioned earlier, the project was implemented in three GHs and five PHCs that were operating in a hub-and-spoke model. Two of the PHCs included in the project covered tribal populations living in forest areas of that district, which were officially designated as backward regions with the lowest Human Developmental Index (0.28) in the district.
As a first step in implementing the project, an educational conference was organized and conducted in conjunction with the local University Hospital. Experts from India, USA, and UK, with extensive experience in high-risk obstetrics as well as simulation training served as faculty at the conference. Various obstetrical emergency causes leading to maternal harm or death, and current treatment guidelines to manage such situations were included in the curriculum. Attendance at the conference was free but mandatory (covered within work hours) for the relevant physicians and nurses from the study hospitals and clinics. This was followed up with trainers from the WONDER team visiting each hospital and PHC and providing group as well as one-on-one training to all the providers at the pilot sites.
The WONDER telehealth project was positioned as a supplement to the current processes in the GH and PHC. To ensure that the project did not disrupt or interfere with existing established hospital and PHC care processes, a small team of hardware, software, and medical technician staff was established (the WONDER project staff) to operate alongside and in parallel with hospital and clinic staff. The implementation and maintenance of the WONDER hardware and software and all data entry in the WONDER system were managed by this separate workforce. The WONDER project staff and supervisors received hospital access clearance from the hospital administration and remained on the hospital premises for the duration of the project. All WONDER staff were trained on maintaining confidentiality of patient information, and all members who provided patient care signed a confidentiality agreement.
WONDER project medical staff were trained and approved to check the VS of all the patients admitted to the labor and delivery area periodically using monitoring kits readily available in India (e.g., digital blood pressure apparatus, thermometer, and pulse oximeters), and to enter the readings into the EHR record for each patient in the WONDER system. The WONDER algorithm continuously scored each patient, and the results were presented on a large dashboard that was placed in a highly visible area adjacent to the nursing station. Visual and audible alerts were created according to the preset criteria by the WONDER system (Table 1). WONDER staff were instructed to check patient’s VS every 4 hours during labor. Any patient identified as having a red alert was monitored every 15 minutes or as directed by the physician. This monitoring continued until the vitals were scored as yellow or green. Since patients who presented with normal VS during labor could develop eclampsia during the postpartum period, VS monitoring continued until each patient was discharged from the hospital. Alerts continued to be generated as long as the scoring values were abnormal.
In the GH, for all women in labor and in postpartum, the WONDER staff also monitored the alerts and personally notified the nursing staff for taking further action for all red alerts. Treatment protocols built into the WONDER system were printed by the WONDER staff and placed on the patient’s charts to help the nurses and physicians to make treatment changes quickly if necessary. In addition, as the system was scoring and generating alerts for all patients currently in labor in various PHCs, the same alerts were visible on the dashboard at the GH. As each alert came in from a satellite PHC, WONDER staff notified the skilled provider in the labor ward who, in turn, communicated with the nurse in the PHC facility to stabilize and transfer the patient to the GH if the clinical situation could not be brought under control in the remote location. As a result, the system was able to provide the nursing and medical staff at the GH with sufficient time to prepare for the patient’s arrival.
During this study, a total of 15,184 patients were monitored, with the majority of patients recorded in the Erode GH (57.3%, 8,695/15,184). The remaining patients were monitored in Gobichettypalayam (15.3%, 2,326/15,184), Chithode (11.9%, 1,800/15,184), Ammapettai (6.8%, 1,030/15,184), Thalavadi (4.6%, 703/15,184), Modakurichy (3.6%, 545/15,184), Sathyamangalam (0.4%, 65/15,184), and Hongalvadi (0.1%, 20/15,184). It is important to note that in Erode, Gobichettipalayam, and Sathyamangalam, there are GHs with full facilities, while the remaining are PHCs, which are more resource limited.
Patient vitals were evaluated at least every 4 hours from first presentation in a healthcare facility until childbirth and discharge, and the patient’s risk of complications was continually assessed by the WONDER system. Of the 15,184 patients examined, 44.1% (6,693/15,184) of them went from initial visit to childbirth, postpartum and discharge with no alerts, meaning that the WONDER telehealth system never identified these women as being at risk of complications (Table 2). None of the patients in this category faced any complications or mortality.
Table 2. Percentage of patients with no alerts, yellow alerts, and red alerts based on the WONDER system
| Facility | Total patients | Patients without alerts (%) | Patients with alerts (%) | Patients with yellow alerts (%) | Patients with red alerts (%) |
|---|---|---|---|---|---|
| Ammapettai | 1,030 | 37.7 | 62.3 | 50.9 | 11.5 |
| Chithode | 1,800 | 39.9 | 60.1 | 38.8 | 21.3 |
| Erode GH | 8,695 | 39.7 | 60.3 | 36.3 | 24 |
| Gobichettypalayam | 2,326 | 69.2 | 30.8 | 20.2 | 10.6 |
| Hongalvadi | 20 | 70 | 30 | 10 | 20 |
| Modakurichy | 545 | 44.6 | 55.4 | 39.3 | 16.1 |
| Sathyamangalam | 65 | 70.8 | 29.2 | 21.5 | 7.7 |
| Thalavadi | 703 | 32.1 | 67.9 | 38.1 | 29.7 |
| Total | 15,184 | 44.1 | 55.9 | 35.2 | 20.7 |
GH, general hospital.
Alerts were issued for 55.9% of women (8,491/15,184). Of these patients, 63.0% (5,346/8,491) of them received only yellow alerts, while 37.0% (3,145/8,491) received red alerts (Figure 2). For the purposes of this analysis, a patient with both red and yellow alerts would be counted in the red alert category. All maternal deaths that occurred over the course of the study resulted from the deteriorating condition of women who received red alerts (20.7%, 3,145/15,184), revalidating the belief that a small number of women with preventable and identifiable problems account for the most severe situations. Approximately 37% of the red alerts came from 21% of patients. This is because women under worsening conditions were more likely to have multiple problems. For instance, a patient experiencing preeclampsia may have had postpartum hemorrhage or subsequent infections, giving rise to multiple red alerts.
Figure 2—Percentage of patients with green, yellow, and red alerts.
Fundamental to the success of WONDER system is that red and yellow alerts garner the necessary medical attention for a given patient, prompting the patient’s stabilization. To this effect, we assessed the number of alerts required to return patient vitals to within normal limits. The results of this study revealed that 66.4% (5,634/8,491) of patients were returned to a healthy state with less than three alerts (Figure 3). However, for 25.9% (2,202/8,491) of patients returning vitals to normal parameters took 4–10 alerts, and for 7.7% (655/8,491) of patients, more than 10 alerts were required. These 7.7% of patients account for nearly a quarter of the total number of alerts issued, suggesting that identifying and addressing issues early on in high-risk patients would mitigate many problems later on. The delay in stabilization for the patients who required more than three alerts could have resulted either from ineffective treatment or from delay in initiating treatment, which highlights an important area for future improvement. An example of a patient with multiple red alerts issued that were not attended to and resulted in a fatality is presented in Figure S1.
Figure 3—Number of alerts required to return to patient vitals within normal limits.
Figure 4 shows the time elapsed between the signaling of a WONDER alert and the initiation of care. This response-time surveillance was instituted during the final 4 months of the study, and analysis was conducted upon the 153 patients included who generated 714 alerts. For 92.2% of these patients (141/153), care was initiated within an hour of the signaling of a red alert. These patients accounted for 95.5% (682/714) of alerts, meaning that the large majority of alerts resulted in treatment of a potentially life-threating problem within 1 hour.
Figure 4—Treatment response time.
This finding provides early evidence that even resource-constrained hospitals are in a position to raise their response times when provided with a clear understanding of what action is necessary and when. However, 7.8% (12/153) of patients waited for more than an hour for treatment, highlighting that there is still room for improvement.
A key finding of this pilot study in TN was a meaningful reduction in MMR. During the 2-year study period, a total of six maternal deaths were reported at the study sites. One of those deaths occurred in post-partum after the patient was discharged, and WONDER monitoring had concluded. This patient was readmitted 3 weeks later in septic shock in a moribund state and went into cardiac arrest in the Emergency Room (ER). The WONDER system identified the risk and issued a red alert with every set of vitals recorded from readmission to death. This alerted the WONDER staff of the urgent situation, who relayed the information to the providers. However, the physicians were not adequately trained to treat the complications that arose during the postpartum period such as deep vein thrombosis and pulmonary embolism. Subsequent to this incidence of maternal death, the State MMR Committee identified the knowledge gap and created treatment guidelines for thromboembolism, which were made available to all providers in the state. This alone demonstrates WONDER’s ability to help identify the knowledge gaps and improve provider’s education. These findings also demonstrate the importance of continued post-partum monitoring even after hospital discharge, a key issue that we intend to address in future studies by extending WONDER monitoring into the postpartum phase. However, for the purpose of our analysis, the first patient was excluded as it did not occur under the supervision of the WONDER telehealth system. Therefore, the total number of deaths reported under WONDER monitoring was 5.
The MMR in TN during the 2 years prior to the implementation of WONDER was 66 per 100,000 live births, or 0.066%, per India’s 2018 State Health Management Information System data.21,22 These data were used as a baseline to compare with maternal mortality rates after WONDER was implemented (Figure 5). During the study period, the MMR in the monitored population declined from 0.066% to 0.03% (5/15,184). While the study period numbers are small, prima facie, they demonstrate a 50.1% reduction in cumulative maternal mortality and evidence the WONDER system’s ability to impact maternal mortality in a short period of time.
Figure 5—MMR reduction in Tamil Nadu during WONDER. *MMR before WONDER as per 2018 State Health Management Information System data. MMR, maternal mortality ratio; WONDER, Women’s Obstetrical Neonatal Death and Reduction telehealth system.
The main outcome studied in this pilot project was the prevalence of in-hospital eclampsia, a leading cause of maternal mortality. Over the course of the study, 9.6% (1,454/15,184) of women developed mild preeclampsia, 3.8% (580/15,184) developed severe preeclampsia, and 27 patients developed eclampsia for a cumulative incidence of 0.18% (27/15,184). When compared with the incidence of eclampsia before WONDER as per 2014 National Eclampsia Registry Data (2.18%), there is a 91.7% decrease in the incidence of eclampsia (Figure 6).18
Figure 6—Incidence of eclampsia reduction in Tamil Nadu during WONDER. *Incidence of eclampsia before WONDER as per 2014 National Eclampsia Registry Data. WONDER, Women’s Obstetrical Neonatal Death and Reduction.
This alone is a very promising finding. However, of the 27 patients who developed eclampsia during the course of this study, 70.3% (19/27) experienced eclampsia at home or in other hospitals where they were outside of monitoring by the WONDER system. Examining the occurrence of eclampsia when under WONDER system monitoring, we found only eight patients developed eclampsia after being admitted to the hospital (0.05%, 8/15,184). We suspect that this extremely low incidence results from the fact that although patients with severe preeclampsia have a high potential to develop eclampsia, under WONDER monitoring, cases of severe preeclampsia were identified early and appropriate treatment was initiated without delay.
This study only implemented WONDER monitoring in the hospital setting. However, 68.4% (13/19) of out-of-hospital eclampsia occurred at home, emphasizing the need for extending WONDER monitoring into the antenatal period, as well as the need for educating patients regarding the warning signs. The remaining 31.6% (6/19) of patients reported eclampsia at health centers or at smaller private hospitals where the providers were not comfortable treating such patients with severe preeclampsia. This highlights the opportunity to expand the coverage of WONDER system to smaller hospitals and help providers to appropriately identify and manage the patients with severe preeclampsia early, thus maximizing the potential to prevent progression to eclampsia.
Several studies, including the United Kingdom’s Confidential Enquiries into Maternal Deaths, indicate that early identification and immediate treatment of high-risk situations without delay are key to preventing maternal deaths.9 WONDER is specifically designed to fill that need and has provided early evidence to its ability to not only identify serious clinical situations as soon as the clinical condition changes but also help in initiating appropriate care using its decision support system. This is made clear by the finding that appropriate treatment was initiated within 1 hour for 92.2% of patients under WONDER monitoring who met the recommended ACOG guidelines for emergent maternal therapy.23 This response time was achieved in a low-resource environment using off-the-shelf biometric devices and low-level providers to help implement care.
By scoring patient conditions (red, yellow, or green), WONDER was able to efficiently allocate available resources to optimize quality of care for all patients. This alert system identified 44.1% of the patients as having no risk factors, allowing those patients to be cared for by low-level providers. For the remaining 55.9% of patients, WONDER directed the care of skilled providers to the sickest patients who most urgently needed advanced care. Both the quick average response time and relatively low average number of alerts required to return critical vitals to within normal limits speak of WONDER’s success in allocating resources. In addition, given the limited resources available in and around the WONDER study sites, these findings suggest that quick response time is possible even in the absence of higher quality facilities.
Using this system, maternal deaths were reduced by 50.1% and in-hospital eclampsia was reduced by 91.7%. These measures are even more meaningful given that WONDER was implemented in a low resources area, without disrupting the existing healthcare system. As a result, WONDER offers a solution that can be effectively implemented in additional resource-constricted areas, providing a solution for countries who are striving to achieve the SDGs for maternal mortality. The system also identified areas of weakness and highlighted opportunities for improvement, namely, morbidities and mortalities that originated in the antenatal and postpartum phases. While the WONDER trial outlined in this study focused only on patients in labor, we are hoping to address this in an expanded trial providing antenatal care using low-level providers is presently in progress in Bihar in India with over 60,000 pregnant women.
There are some limitations to the use of the WONDER system. The initial cost of hardware, personnel, and training may be expensive in some instances, although once installed maintenance is minimal. In addition, WONDER requires trained personnel at the electronic birthing centers or tertiary care hospitals to monitor patients from all connected PHCs and Level I hospitals. Computer literacy, broadband connectivity, time, and personnel pose added limitations. An automated system to upload the vitals could mitigate personnel and time limitations. Overall, the system provides a new approach to maternal mortality reduction, which utilizes technology to more effectively monitor and provide care for women in resource-constricted areas of the world.
The authors thank all of their colleagues in Tamil Nadu who helped to implement and use WONDER, and the resilient mothers of the community who have been willing to use this new system.
Funding statement: This work was funded by grants from HP Inc. and from the Erodu Olirum Foundation. The authors acknowledge that HP Inc. and the Erodu Olirum Foundation had no role in study design, implementation, data collection, analysis, decision to publish, or in the preparation of the manuscript.
Conflicts of Interest: The corresponding author, Dr. Narmadha Kuppuswami, of this research study is the CEO of WONDER. The remaining authors declare no potential conflicts of interest.
Contributors: All authors have reviewed, discussed, and agreed to their individual contributions and roles.
| 1. | World Health Organization. Maternal mortality facts sheet. 2019 [cited 2020 Dec 16]. Available from: https://www.who.int/news-room/fact-sheets/detail/maternal-mortality |
| 2. | Stanton ME, Kwast BE, Shaver T, McCallon B, Koblinsky M. Beyond the safe motherhood initiative: Accelerated action urgently needed to end preventable maternal mortality. Glob Health Sci Pract. 2018;6(3):408–12. https://doi.org/10.9745/GHSP-D-18-00100 |
| 3. | Alkema L, Chou D, Hogan D, et al. Global, regional, and national levels and trends in maternal mortality between 1990 and 2015, with scenario-based projections to 2030: A systematic analysis by the UN Maternal Mortality Estimation Inter-Agency Group. Lancet. 2015;387(10017):462–74. https://doi.org/10.1016/S0140-6736(15)00838-7 |
| 4. | United Nations Department of Economic and Social Affairs, Statistics Divisions. The sustainable development goals report 2020. [cited 2020 Dec 16]. Available from: https://unstats.un.org/sdgs/report/2020/ |
| 5. | United Nations. The millennium development goals report 2015. [cited 2020 Dec 2016]. Available from: https://www.un.org/millenniumgoals/2015_MDG_Report/pdf/MDG%202015%20rev%20(July%201).pdf |
| 6. | World Health Organization. India has achieved groundbreaking success in reducing maternal mortality. June 10, 2018 [cited 2020 Dec 16]. Available from: https://www.who.int/southeastasia/news/detail/10-06-2018-india-has-achieved-groundbreaking-success-in-reducing-maternal-mortality |
| 7. | Kanti V, Sharma P, Seth S. Maternal mortality trends in Uttar Pradesh, India. J South Asian Feder Obstet Gynecol. 2018;9(4):372–6. https://doi.org/10.5005/jp-journals-10006-1532 |
| 8. | Mukherjee S, Mukherjee S, Sarkar R. A six year retrospective study of maternal mortality at a tertiary teaching institute in Uttarpradesh. Int J Med Sci Public Health. 2014;3(11):1407–9. https://doi.org/10.5455/ijmsph.2014.220820142 |
| 9. | Barnes-Josiah D, Myntti C, Augustin A. The “three delays” as a framework for examining maternal mortality in Haiti. Soc Sci Med. 1998 Apr;46(8):981–93. https://doi.org/10.1016/s0277-9536(97)10018-1 |
| 10. | Pacagnella RC, Cecatti JG, Osis MJ, Souza JP. The role of delays in severe maternal morbidity and mortality: Expanding the conceptual framework. Reprod Health Matters. 2012;20(39):155–63. https://doi.org/10.1016/S0968-8080(12)39601-8 |
| 11. | Chavane LA, Bailey P, Loquiha O, et al. Maternal death and delays in accessing emergency obstetric care in Mozambique. BMC Pregnancy Childbirth. 2018;18:71. https://doi.org/10.1186/s12884-018-1699-z |
| 12. | Salam RA, Das JK, Ali A, Bhaumik S, Lassi ZS. Diagnosis and management of preeclampsia in community settings in low and middle-income countries. J Fam Med Prim Care. 2015;4(4):501–6. https://doi.org/10.4103/2249-4863.174265 |
| 13. | Shennan AH, Redman C, Cooper C, Milne F. Are most maternal deaths from pre-eclampsia avoidable? Lancet. 2012;379(9827):1686–7. https://doi.org/10.1016/s0140-6736(11)60785-x |
| 14. | ACOG Committee on Practice Bulletins—Obstetrics. ACOG practice bulletin. Diagnosis and management of preeclampsia and eclampsia. Number 33, January 2002. Obstet Gynecol. 2002 Jan;99(1):159–67. https://doi.org/10.1016/s0029-7844(01)01747-1 |
| 15. | Gupte S, Wagh G. Preeclampsia-eclampsia. J Obstet Gynaecol India. 2014 Feb;64(1):4–13. https://doi.org/10.1007/s13224-014-0502-y |
| 16. | Abalos E, Cuesta C, Grosso AL, Chou D, Say L. Global and regional estimates of preeclampsia and eclampsia: A systematic review. Eur J Obstet Gynecol Reprod Biol. 2013 Sep;170(1):1–7. https://doi.org/10.1016/j.ejogrb.2013.05.005 |
| 17. | American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics. Gestational hypertension and preeclampsia: ACOG practice bulletin, Number 222. Obstet Gynecol. 2020 Jun;135(6):e237–60. https://doi.org/10.1097/AOG.0000000000003891 |
| 18. | Nobis PN, Hajong A. Eclampsia in India through the decades. J Obstet Gynaecol India. 2016;66(1):172–6. https://doi.org/10.1007/s13224-015-0807-5 |
| 19. | Vousden N, Lawley E, Seed PT, et al. Incidence of eclampsia and related complications across 10 low- and middle-resource geographical regions: Secondary analysis of a cluster randomised controlled trial. PLoS Med. 2019;16(3):e1002775. https://doi.org/10.1371/journal.pmed.1002775 |
| 20. | Nour NM. An introduction to maternal mortality. Rev Obstet Gynecol. 2008;1(2):77–81. |
| 21. | Ministry of Health and Family Welfare (India). India HMIS standard report—Data itemwise monthly 2017–2018. New Delhi, India: Ministry of Health and Family Welfare (India). 2018. |
| 22. | Josephine SM. Tamil Nadu’s maternal mortality rate sees sharp six-point drop. The Hindu. July 2019. Available from: https://www.thehindu.com/news/national/tamil-nadu/states-mmr-sees-sharp-six-point-drop/article28529982.ece |
| 23. | ACOG Committee on Obstetric Practice. ACOG Committee Opinion. Emergent therapy for acute-onset, severe hypertension during pregnancy and the postpartum period. Number 767, February 2019. Available from: https://www.acog.org/clinical/clinical-guidance/committee-opinion/articles/2019/02/emergent-therapy-for-acute-onset-severe-hypertension-during-pregnancy-and-the-postpartum-period |
Copyright Ownership: This is an open access article distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, and enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See http://creativecommons.org/licenses/by-nc/4.0.