ORIGINAL RESEARCH
Strategies to Improve Time to Activation Within an Ambulatory Remote Patient Monitoring Program
Mark R. Stemler, MHA1 , Nicole J. Ploog, MBA, MS2
, Nicole J. Ploog, MBA, MS2 , Shelby R. Gathje, MHA3
, Shelby R. Gathje, MHA3 and Jordan D. Coffey, MBA, MHA, MA2
and Jordan D. Coffey, MBA, MHA, MA2
1Strategy Department, Mayo Clinic, Rochester, MN, USA; 2Center for Digital Health, Mayo Clinic, Rochester, MN, USA; 3Research Administration, Mayo Clinic, Rochester, MN, USA
Abstract
Remote patient monitoring (RPM) programs decrease rates of healthcare utilization among patients with chronic conditions. Immediately enrolling a patient and activating them in the RPM program, either upon or soon after discharge, is an important step in achieving these benefits. We tested interventions across three Plan-Do-Study-Act quality improvement cycles to understand the extent to which operational improvements lead to timely activation. Each improvement cycle resulted in decreased time to activation, with the cumulative effect—as applied to patients on the COVID-19 RPM program—resulting in a reduction that was overall greater than the sum of the individual improvements. As additional healthcare systems develop and deploy RPM programs, the learnings from this project can provide insight into the operational and logistical challenges encountered in providing these services, along with potential interventions to achieve timely activation.
Keywords: care coordination, COVID-19, process optimization, quality improvement, remote patient monitoring, RPM strategies
Citation: Telehealth and Medicine Today 2022, 7: 361 - http://dx.doi.org/10.30953/tmt.v7.361
Copyright: © 2022 The Authors. This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0.
Received: March 18, 2022; Accepted: March 31, 2022; Published: May 10, 2022
Funding Statement: There are no funding sources that need to be disclosed.
Financial and non-Financial Relationship and Activities: The authors declare no potential conflicts of interest.
Correspondence: Mark R. Stemler, Email: Stemler.mark@mayo.edu
In 2016, there were 49.22 million U.S. citizens over the age of 65 years.1 By 2030, that number is projected to increase to 74 million.2 As the aging population increases, so too will the number of individuals with chronic diseases, including heart failure, diabetes, hypertension, and respiratory conditions.3,4 In response to this rising number of patients, as well as additional challenges related to limited hospital capacity and shortages of healthcare staff,5 many healthcare organizations are developing and refining transitional care programs.4,6
Transition to an appropriate ambulatory treatment plan is vital for reducing the rate of hospitalization among chronically ill patients.7 Ambulatory remote patient monitoring (RPM) programs have effectively reduced avoidable emergency department visits and hospital admissions.8,9 Since 2016, our organization—a large, geographically dispersed academic medical center and associated health system—has deployed a robust RPM program as an essential strategy to care for chronically and acutely ill patients through advanced digital connection opportunities that complement integrated care delivery.10 This program utilizes medical-grade equipment deployed to patients’ homes to provide health data to a centralized team of overseeing clinical professionals.11
To receive the full benefits of an RPM program, it is critical to not only deploy equipment to enrolled patients but efficiently move them to an active state on the program as rapidly as possible.9 Nearly one-third of hospital readmissions occur within 1 week of discharge.12 Further, prior studies show that the effective transition of patients to suitable posthospital care programs in fewer than 10 days significantly reduces the rate of hospital readmissions.13,14
We undertook Plan-Do-Study-Act (PDSA)15 quality improvement cycles to decrease the time between a patient’s hospital discharge and when they became active in the RPM program. This paper discusses the methods, population, and approaches to achieve these goals, as well as associated measures and results. Finally, we review the lessons learned, considerations for other healthcare organizations, and overall limitations of this evaluation.
Materials and Methods
The end-to-end RPM value stream was analyzed to determine how to achieve patient activation within a 10-day window. Four distinct phases in the patient journey were identified: patient qualification, equipment transit (i.e., logistics), activation (i.e., submission of first vital signs), and ongoing monitoring. Among patients enrolled in RPM, only 40% achieved activation by day 10. This led the team to analyze improvement opportunities across the first three phases of the RPM value stream.
Stakeholder input was garnered through a multidisciplinary team, including nursing, allied health staff, administration, and a third-party logistics partner. Stakeholders participated in cause-and-effect analysis and brainstorming sessions to ideate possible alternative solutions to the gap in quality.
This team met regularly to strategize and ensure adherence to clinical practice standards and vendor partner contract stipulations. In addition, patient experience experts were consulted to validate the impact of proposed improvement efforts on the patient experience. Through stakeholder feedback, quality improvement methodology, literature review, and evaluation of first-time quality, and wait time delays, it was determined that the greatest opportunity for improvement fell within the activation phase of the patient journey. Therefore, the goal of the improvement initiatives was to achieve 80% activation in less than 10 days postdischarge. To achieve this goal, a PDSA framework was selected as the vehicle to test quickly, observe results, and act on learnings. Three specific interventions were selected: (1) improved patient experience through streamlined packaging, instructions, and messaging; (2) improved logistics; and (3) patient engagement initiation and its impact on activation.
Interventions
Cycle one: Improved packaging, instructions, and messaging
A patient experience study was conducted to understand how patients reacted to the equipment arriving at their home and the process they undertook to unbox and set up the equipment. The team identified opportunities to streamline the packaging, labeling, and organization of RPM equipment and instructions. As a result, the RPM team reduced the instructional material to incorporate visual cues to guide users through unboxing, equipment setup, and submission of first vital signs. Packaging materials were changed from an oversized cardboard “crate” style box with bubble-wrapped peripherals to a cardboard “pizza style” box with a handle and die cut foam housing the peripherals. These changes sought to improve approachability and reinforce the ease of activating via the technology and equipment. To accompany the experience improvements to the box, program staff were given amended welcome call scripts to reinforce the ease of self-installation.
Cycle two: Improved logistics
When the RPM program was established, a signature was required upon delivery of the RPM equipment to help safeguard against loss. This process led to numerous failed deliveries. The second PDSA cycle eliminated the need for a patient signature, tested improvements in third-party vendor weekend shipping practices, and streamlined patient contact processes, all to decrease equipment transit time and remove barriers to delivery.
Cycle three: Patient engagement initiation and its impact on activation
The onset of the COVID-19 pandemic created the need to scale the RPM program rapidly. Over 8 months from March 26 to November 30, 2020, more than 8,500 patients with COVID-19 were enrolled for monitoring.11 To meet the demands of the practice within the institution, a third PDSA cycle was enacted to test overnight shipping at scale, nurse–patient engagement regardless of RPM equipment activation, and engagement of a centralized provider team to facilitate patient enrollment. Additionally, patient support videos, available asynchronously, were created to reinforce program goals and equipment ease of use.
Population
This evaluation represented an unblinded retrospective analysis of patients enrolled in the RPM program as a standard part of their care between May 1, 2019 and October 30, 2020. This project was reviewed by the Mayo Clinic Institutional Review Board and deemed not human subject research. Patients enrolled in RPM programs included those with chronic, acute, or subacute conditions who were otherwise at risk for hospitalization and were enrolled from either the ambulatory or inpatient settings for outpatient monitoring. Standard program inclusion criteria specified that patients be community dwelling of 18 years old or greater; have a qualified overseeing provider; overall willing to actively engage in their care; and not otherwise have physical, mental health, or uncontrolled substance abuse issues that would limit their ability to participate independently in the RPM program. It was imperative to ensure that process changes to improve activation presented no additional barriers to willing patient participation or cause them to cancel their participation. Cancelation rates served as the balancing measure to assess potential negative impacts of the interventions on the patient.
A convenience sample of 4,233 participants was selected from among the overall program participants following the intention-to-treat analysis principle. A post hoc power analysis was conducted using G*Power software (version 3.1.9.7) based on the average time to activation and standard deviation for each pairing. A one-tailed Wilcoxon–Mann–Whitney test of two groups was utilized with an a of 0.05 and applied to the participant data. This analysis identified a small relative effect size (Cohen’s d = 0.233) for the analysis of cycle one relative to the baseline with an achieved power of 90.55%; a large relative effect size (Cohen’s d = 0.7496 and 1.6699, respectively) for the analysis of cycles two and three each relative to baseline with an achieved power of 100%; a medium and large relative effect size (Cohen’s d = 0.5655 and 1.6226), respectively, for the analysis of cycles two and three each relative to cycle one with an achieved power of 100%; and a large relative effect size (Cohen’s d = 0.9719) for the analysis of the cycle two relative to cycle three with an achieved power of 100%.
Measures
Time to activation within each quality improvement cycle was measured from the point the RPM program was ordered by the frontline clinical team to the point in which the patient returned one or more electronic vital signs through the RPM electronic platform. Data were collected from the electronic health record and patient delivery and install statistics collected from the logistics partner. Patient RPM program completion was based on patients reaching desired clinical goals as documented by nurse assessment or patient exiting the program before reaching this endpoint. Patient disposition (i.e., program enrollment while classified as inpatient status or otherwise while outpatient status) was abstracted from the electronic medical record by program staff.
Statistical Analysis Approach
Primary outcome analyses compared pairwise mean time to activation between each cohort. All statistical tests were conducted with BlueSky Statistics (Commercial Server Edition version 7.20). Normality was assessed utilizing the Shapiro Wilk test (p = 0.00). A log transformation was attempted but did not allow for normal distribution. Overall pairings were analyzed via nonparametric Kruskal–Wallis rank sum tests. Subsequent pairwise analyses of effects were evaluated via two-sided Wilcoxon rank sum tests. As noted, participant completion was analyzed with an intention-to-treat analysis, with participants lost to follow-up assumed to have otherwise canceled from the program.
Results
There were 491 participants included in the baseline group: 257 participants included in the cycle one cohort, 388 participants in the cycle two cohort, and 3,097 participants in the third cycle cohort (Table 1). In total, 40% of baseline participants reached activation within 10 days following the initial RPM program order. This activation rate rose to 55% among participants who were in cycle one, 80% among participants who were in cycle two, and 98% among participants who were in cycle three (Table 1). Cancelation rates were not different (Table 1).
Significant (p < 0.05) improvements in time to activation were observed among all pairings (baseline to cycle one, baseline to cycle two, baseline to cycle three, cycle one to cycle two, cycle one to cycle three, and cycle two to cycle three) (Table 2, Figure 1). Time to activation differed significantly (p < 0.05) between inpatient and outpatient referral within each intervention. Given these differences, a subgroup analysis was conducted within each intervention pairing to evaluate any effect modification that may result from the referral source. Improvements remained significant (p < 0.05) upon subgroup analysis of within each pairing (Table 3, Figure 2).
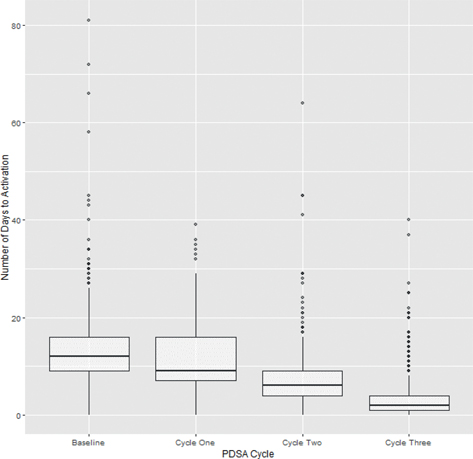
Fig. 1 Comparison of days to patient activation between Plan-Do-Study-Act (PDSA) cycles
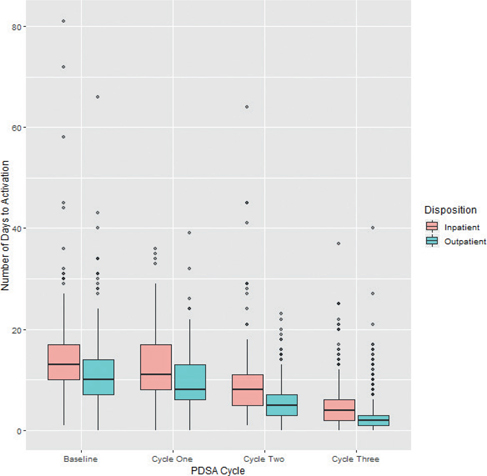
Fig. 2 Subgroup analysis—days to patient activation between Plan-Do-Study-Act (PDSA) cycles taking into account whether the patient was enrolled to remote patient monitoring (RPM) while hospitalized or postdischarge (in the ambulatory environment)
Discussion
The results suggest that methods to simplify the patient experience around receiving and engaging with at-home clinical support equipment, as well as ongoing engagement with the care team while logistics are underway, significantly reduce the time between patient enrollment and active participation in RPM. Although differences in time to activation were observed for those referred while inpatient versus those referred while outpatient, these differences were expected given that enrollment of patients from the hospital setting often occurs prior to discharge, creating a lag in activation that results from the hospitalized period. Although this study did not directly track the patient experience, we did not observe an increase in program cancelation rates, which we believe to be indicative that the patient experience was not otherwise negatively impacted.
To help reduce the transmission of the COVID-19 virus, approaches to provide effective care outside traditional brick and mortar medical facilities have become increasingly important.16 This shift has resulted in patients and care teams alike embracing digital modalities of care.11 As these digitally based approaches continue to advance, organizations must apply a thoughtful, intentional approach to engage patients successfully in the ambulatory setting.
When the RPM program began at our healthcare organization, care teams were hesitant to take any part in patient identification, equipment distribution, or active monitoring of patients. By embracing ongoing quality improvement and process optimization, the team was able to achieve successful engagement, which allowed the RPM program to scale with agility in response to the COVID-19 pandemic.
Considerations for Other Healthcare Systems
Patient focus is a key foundational component of any successful clinical program, especially in the digital space where it can be difficult to create and maintain meaningful connections with patients.17 The RPM program accomplishes this by first creating a connection in the inpatient environment and then extending it into patient postdischarge programs. The team discovered that care plan initiation by the RPM registered nurse prior to submission of first vital signs was a valuable tool in gaining patient engagement with the overall monitoring plan. Patients knew what to expect because of the touchpoint they had with their RPM registered nurse and improved confidence when interacting with potentially confusing technology. This created a collaborative approach where both the patient and the registered nurse took active roles in monitoring and improving the patient’s health.
Patient experience must be considered when developing systems to deliver RPM solutions. Experience design experts conducted field interviews and observations of patients interacting with the packaging and engaging in equipment setup, which provided valuable “voice of the customer” insight into the challenges with the boxed kit and the peripheral set-up process. From the interviews/observations, it was determined that many patients felt overwhelmed and confused by the large volume of information. Given these findings, the team worked with multiple stakeholders across the organization, resulting in streamlined packaging and instructions, which leveraged graphics and videos rather than lengthy written text to communicate. Further, it was determined that RPM equipment should be configured prior to delivery, with all the necessary components connected and the features aligned with the patient’s diagnosis. If the patient must configure the technology themselves, the steps should be as simple as possible, with clear instructions of whom to contact regarding patient care or technology difficulties.
Finally, steps should be taken to make it as easy as possible for patients to receive their equipment. Although there were anecdotal cases of equipment loss following the elimination of the signature requirement for delivery, these data were not readily available for analysis. Based on interviews with staff, equipment loss remained infrequent and unlikely to offset the gains in efficiency associated with the change.
Limitations
This project used a quality improvement methodology with evaluative measures analyzed retrospectively. The team assessed a nonrandomized convenience sample, and no methods were used to systematically account for potential confounding or bias. This methodology made it difficult to definitively determine whether the improvements were causally related to patients activating sooner. Even though this evaluation represented learnings from large patient cohorts across each PDSA cycle, a disproportionate number of patients were present in the PDSA cycle conducted during the COVID-19 pandemic. Although the overall care model was the same regardless of condition, the acute nature of COVID-19 necessitated that care teams engage more frequently during the preactivation process with patients in this program than with patients in the chronic condition programs. This increased engagement may have subsequently impacted activation time in a manner that was not evaluated directly in this analysis. Cancelation rates were used as the sole measure of patient experience; other qualitative factors measuring patient satisfaction or understanding of the program could have also been used to measure the effect.
Although this project was conducted across multiple physical locations and practice settings, it represents learnings from a single organization and may not otherwise be generalizable to other healthcare organizations. Further, the endpoints and analysis focused on the time between enrollment and activation in the RPM program. Additional analysis postactivation is subject to further study. Finally, observations and interviews with patients were conducted in the beginning of the project by experience design experts, but patient representatives were not actively engaged as stakeholders over the remaining course of this project.
Conclusions
Each of the three PDSA cycles improved the activation rates in patients enroled in an ambulatory RPM program. These learnings can provide helpful considerations for identifying and addressing potential operational and logistical challenges as more healthcare organizations develop systems and processes to provide supportive care outside of traditional brick-and-mortar facilities.
Authors’ contributions
All authors approved the manuscript and meaningfully contributed to the submission, thereby adhering to the proper ethics of authorship.
Acknowledgements
The authors acknowledge the contributions of the Mayo Clinic RPM Desk Operations Specialist and nursing teams for incorporating these process changes and providing feedback. Additionally, the authors acknowledge the direct support and contributions of Nina Sargent, Ben Barrett, Jen Soberg, Lukas Manka, Kris Pearson, Lindsay Rhodes, Roxanna Holper, Carole Berteotti, Anne Schwartz, Lori Neu, Nicole Blegen, Sarah Bell, and Tufia Haddad, MD.
References
- Roberts AW, Ogunwole SU, Blakeslee L, Rabe MA. The population 65 years and older in the United States: 2016 [Internet]. Washington, DC: U.S. Census Bureau; 2018 [cited 2021 November 5]. Report No.: ACS-38. Available from: https://www.census.gov/content/dam/Census/library/publications/2018/acs/ACS-38.pdf
- Colby SL, Ortman JM. Projections of the size and composition of the U.S. Population: 2014 to 2060 [Internet]. Washington, DC: U.S. Census Bureau; 2015 [cited 2021 November 5]. Report No.: P25-1143. Available from: https://www.census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf
- Atella V, Piano Mortari A, Kopinska J, Belotti F, Lapi F, Cricelli C, et al. Trends in age-related disease burden and healthcare utilization. Aging Cell [Internet]. 2019 [cited 2021 November 5];18(1):e12861.Available from: https://onlinelibrary.wiley.com/doi/10.1111/acel.12861
- Maresova P, Javanmardi E, Barakovic S, Barakovic Husic J, Tomsone S, Krejcar O, et al. Consequences of chronic diseases and other limitations associated with old age—a scoping review. BMC Public Health. 2019:19;1431. doi: 10.1186/s12889-019-7762-5
- Bazzoli GJ, Brewster LR, Liu G, Kuo S. Does U.S. hospital capacity need to be expanded? Health Aff. 2003:22(6);40–54. doi: 10.1377/hlthaff.22.6.40
- Hansen LO, Young RS, Hinami K, Leung A, Williams MV. Interventions to reduce 30-day rehospitalizations: a systematic review. Ann Intern Med. 2011;155(8):520–8. doi: 10.7326/0003-4819-155-8-201110180-00008
- UpToDate [Internet]. Wolters Klower; c2021. Hospital discharge and readmission [updated 2021 September 30; cited 2021 November 5]. Available from: https://www.uptodate.com/contents/hospital-discharge-and-readmission
- Crossen-Sills J, Toomey I, Doherty M. Strategies to reduce unplanned hospitalizations of home healthcare patients: a step-by-step approach. Home Healthc Nurs. 2006;24(6):36976. doi: 10.1097/00004045-200606000-00007
- Su D, Michaud TL, Estabrooks P, Eiland LA, Schwab RJ, Hansen G, et al. Diabetes management through remote patient monitoring: the importance of patient activation and engagement with the technology. Telemed J E Health. 2019:25(10):952–9. doi: 10.1089/tmj.2018.0205
- Haddad TC, Blegen RN, Prigge JE, Cox DL, Anthony GS, Leak MA, et al. A scalable framework for telehealth: the Mayo Clinic Center for Connected Care response to the COVID-19 pandemic. Telemed Rep. 2021;2(1):78–87. doi: 10.1089/tmr.2020.0032
- Coffey JD, Christopherson LA, Glasgow AE, Pearson KK, Brown JK, Gathje SR, et al. Implementation of a multisite, interdisciplinary remote patient monitoring program for ambulatory management of patients with covid-19. NPJ Digit Med. 2021;4:123. doi: 10.1038/s41746-021-00490-9
- Healthcare Cost and Utilization Project [internet]. Rockville, MD: Agency for Healthcare Research and Quality; 2017. A comparison of all-cause 7-day and 30-day readmissions; [updated 2017 October 31; cited 2021 October 12]. Available from: https://www.hcup-us.ahrq.gov/reports/statbriefs/sb230-7-Day-Versus-30-Day-Readmissions.pdf
- Ponniah A, Shakib S, Doecke CJ, Boyce M, Angley M. Post-discharge medication reviews for patients with heart failure: A pilot study. Pharm World Sci. 2008;30(6):810. doi: 10.1007/s11096-008-9230-7
- Logue MD, Drago J. Evaluation of a modified community based care transitions model to reduce costs and improve outcomes. BMC Geriatr. 2013;13:94. doi: 10.1186/1471-2318-13-94
- Plan-Do-Study-Act (PDSA) directions and examples [Internet]. Rockville, MD: Agency for Healthcare Research and Quality; 2020. Available from: https://www.ahrq.gov/health-literacy/improve/precautions/tool2b.html
- Ting DSW, Carin L, Dzau V, Wong TY. Digital technology and COVID-19. Nat Med. 2020;26:459–61. doi: 10.1038/s41591-020-0824-5
- Bailey JE, Gurgol C, Pan E, Njie S, Emmett S, Gatwood J, et al. Early patient-centered outcomes research experience with the use of telehealth to address disparities: scoping review. J Med Internet Res. 2021;23(12):e28503. doi: 10.2196/28503.
Copyright Ownership: This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0.