ORIGINAL RESEARCH
Remote Patient Monitoring Effectively Assures Continuity of Care in Asthma Patients during the COVID-19 Pandemic
Christopher March, BS1  , Kimberly Gandy, MD, PhD2,3
, Kimberly Gandy, MD, PhD2,3  , Jos Domen, PhD3
, Jos Domen, PhD3  , Sayyed Hamidi, MD, MBA, MPH4
, Sayyed Hamidi, MD, MBA, MPH4  , Ryan Chen, BS5
, Ryan Chen, BS5  , Paul Barach, MD, MPH6,7
, Paul Barach, MD, MPH6,7  , Anthony Szema, MD1,8
, Anthony Szema, MD1,8 
1Three Village Allergy and Immunology, South Setauket, New York; 2Department of Biomedical and Health Informatics, University of Missouri, Kansas City, Kansas City, Missouri, USA; 3Play-it Health, Inc., Overland Park, Kansas, USA; 4University of Illinois at Chicago, U.S. Department of Veterans Affairs, Chicago, Illinois; 5Stony Brook University, Stony Brook, New York, USA; 6Jefferson College of Population Health, Philadelphia, Pennsylvania, USA; 7Interdisciplinary Research Institute for Health Law and Science, Sigmund Freud University Vienna, Vienna, Austria; 8Zucker School of Medicine at Hofstra/Northwell; College of Engineering and Applied Sciences, Stony Brook University, Stony Brook, New York, USA.
Keywords: asthma; coronavirus disease; COVID-19; quality; remote patient monitoring; telehealth
Abstract
Background: Digital health tools to bridge gaps in managing infectious pandemics was a proposition grounded, until recently, more hypothetically than in reality. The last 2 years exposed the extraordinary global need for robust digital solutions.
Objective: Determine the ability of remote patient monitoring (RPM) during the COVID-19 (coronavirus disease) pandemic to improve clinical outcomes and assure continuity of care in patients with asthma.
Methods: A total of 102 patients with asthma were enrolled in a telemonitoring protocol at the beginning of the COVID-19 pandemic in the United States. Intervention combined health coaching telephone calls and remote telemonitoring at a private, university-affiliated, outpatient clinical adult, and pediatric allergy/immunology and pulmonary practice. Patients enrolled with the primary rationale of maintaining continuity of care in the face of uncertain clinical care options. Enrollment and data collection proceeded in a fashion to allow detailed retrospective analysis. Telemonitoring included a pulse oximeter linked to a smartphone using the software platform Plan-it Med (PIM)®. A healthcare professional monitored data daily, and patients were contacted by providers due to vital sign abnormalities and treatment plan alterations. Patients were encouraged to remain on the platform daily during the first 3 months of the pandemic. After respiratory and/or clinical stability was achieved and clinic visit opportunities were resumed, patients were encouraged to maintain engagement with the platform but were not expected to use the platform daily. Asthma control test (ACT) scores were recorded before and after 6 months. Paired Wilcoxon signed-rank tests (dependent groups, before vs. after) and Wilcoxon rank-sum (Mann–Whitney) tests were performed for unpaired results (independent groups, RPM vs. control).
Results: Among 102 patients 19 had physiological abnormalities detected (18.6%). Eight of these 19 patients had actionable changes in prescription regimens based on RPM findings (42.1%). In patients utilizing RPM, there was a reported decrease in shortness of breath episodes and decreased need for rescue inhalers/nebulizer medications (P = 0.005). Daily engagement in the first 3 months of the protocol was 61%. Subset analysis revealed 48 study participants (47.1%) chose to continue to actively use the program for at least 14 months. RPM patients (n = 54) were 99.1% compliant with RPM after 110 patient months. Among patients who discontinued the RPM program, reasons included: 1) symptom alleviation (41.7%), 2) out-of-pocket costs to patients (38.9%), and 3) difficulty using the RPM program (16.7%).
Conclusions: Telemedicine is a valuable adjunct to face-to-face visits for asthma care. A novel RPM technology positively impacted continuity of care, asthma outcomes, quality of life, and self-care. Remote therapeutic monitoring offers great promise as a diagnostic tool and therapeutic intervention to improve adherence for patients with difficult-to-control asthma.
Citation: Telehealth and Medicine Today © 2022, 7: 374 - http://dx.doi.org/10.30953/tmt.v7.374
Copyright: © 2022 The Authors. This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0.
Received: 3 October 2022; Revised: 10 October 2022; Accepted: 18 October 2022; Published: 17 November 2022
Financial and non-Financial Relationship and Activities: The authors report no funding for preparation of this article.
Mr. March reports no relevant financial or nonfinancial competing interests to report. Dr. Gandy and Dr. Domen are employed by Play-it Health. Dr. Barach advises Play-it Health. Anthony Szema is Managing Member, Three Village Allergy & Asthma, PLLC; Co-investigator, Columbia University Global Psychiatric Epidemiology Group on CDC NIOSH U01 ‘9/11 Trauma and Toxicity: Longitudinal Health and Behavioral Outcomes’ (PI Christina Hoven); consultant on NIH R01 HL152385, (Hoven, Larry Amsel (MPIs) ‘Childhood Mass Trauma Exposure, Inflammatory Programming, and Psychopathology in Young Adulthood’; Principal Investigator; Principal Investigator NY State A Manufacturing and Technology Resource Consortium (MTRC) Supplemental Award Agreement MTRC Project No.: 20211012.1605; Principal Investigator Butterfly ultrasound storage grant for resource-limited study. The authors declare no external funding for this study.
Corresponding Author: *Paul Barach, Email: paul.barach@jefferson.edu
Infectious pandemics have re-emerged as dominant threats to public health. Though population health experts warned about upcoming pandemics for decades, national and global preparedness for COVID-19 (coronavirus disease) pandemic was limited (1, 2). Digital health tools including telehealth and telemonitoring could prove vital in these scenarios, but they have yet to reach maturity or implementation at scale. Few groups were prepared to launch telemonitoring of patients at the beginning of the COVID-19 pandemic. Countries that were able to mobilize and utilize digital health tools in organized and directed manners faired the best in the pandemic to date (3).
Telemonitoring (remote patient monitoring [RPM]) allows patients to record and send biometric data between clinical visits to their providers through devices (i.e., scales, blood pressure cuffs, oximeters, glucometers, watches, wristbands, skin patches, textiles, smartphones). These data can be evaluated in real time or during clinical visits (4, 5). The recorded data provide insights into patient conditions and experiences allowing efficient prevention, diagnosis, and treatment (6). RPM has been important during the COVID-19 pandemic to optimize healthcare delivery to patients while maintaining healthcare worker safety and assuring care continuity (7, 8). These extra health-oriented touchpoints can improve digital health literacy (9).
The benefits of RPM are well documented in patients with type 2 diabetes mellitus and heart failure. A 2019 study found that diabetic patients who used RPM saw a decrease in HbA1c levels (10), and multiple meta-analyses have demonstrated the potential for RPM as an effective tool for monitoring and controlling HbA1c levels in diabetic patients (11–16). The incorporation of RPM alongside standard patient care for patients suffering from chronic heart failure has demonstrated a significant reduction in the number of patient hospitalizations (17–19), patient morbidity, and mortality rates (20).
The benefits of remote monitoring technology for patients suffering from acute and chronic dyspnea related to asthma, however, have not been convincing. There is limited evidence that RPM use improves the quality of life (HRQoL) (21, 22) for COPD patients, and similarly, there is moderate to strong evidence of its ability to significantly reduce the number of hospitalizations (21–24). Two studies identified correlations between recorded vital signs and the exacerbation of COPD symptoms: one through the interpretation of pulse rate, oxygen saturation, and respiratory rate (25) and the other through recorded FVC and FEV1 values (26). A 2020 study found that asthma patients who received reminders and feedback via RPM regarding the need for inhaled corticosteroid (IC) and short-acting-beta2-agonist (SABA) use required less inhaler use while maintaining IC adherence (27). A 2016 meta-analysis of 18 studies comprising 2,268 participants, however, was unable to show whether asthma telemonitoring with feedback from a healthcare professional increased or decreased the odds of exacerbation of a patient’s symptoms requiring intervention (such as a course of oral steroids or the involvement of emergency services) compared with traditional care methods (28).
The aim of the study was to evaluate the efficacy of RPM for maintaining continuity of care in asthmatic patients with acute and chronic dyspnea in the early days of the COVID-19 pandemic. Additionally, we sought to determine whether RPM could prevent exacerbations of pulmonary pathophysiology and improve clinical outcomes.
Methods
Setting and Participants
We examined a cohort of patients from an outpatient clinical adult and pediatric allergy, immunology, and pulmonology practice in Long Island, New York, USA (Three Village Allergy and Asthma, PLLC, South Setauket, New York, USA), during the height of the COVID-19 pandemic (February 3, 2020 to April 30, 2021). Patients with asthma who had the ability to provide voluntary consent, the ability to comprehend written English, and access to a smartphone/tablet with an associated cellular or Wi-Fi signal were included in this study. We excluded patients unable to provide consent, to read English, and those lacking access to a smartphone/tablet with an associated cellular or Wi-Fi signal.
Study Design
We began organizing RPM beginning in December 2019. The study protocol was approved by the clinical practice’s research committee and patients provided written informed consent at the time of enrollment. The team discussed and reviewed all concerns and confirmed understanding prior to consent. Subjects were required to reaffirm their full consent by an app messaging, texting, phone call, and/or video conferencing, when they downloaded the mobile application for RPM monitoring (Fig. 1).
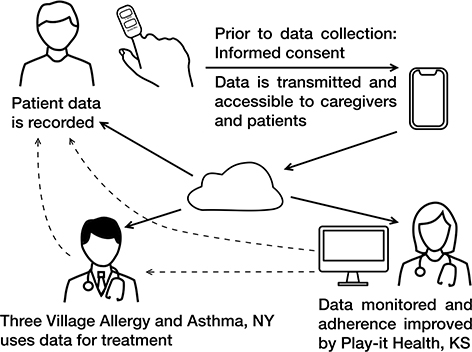
Fig. 1. Diagram describing the transfer of patient data used in the study. Solid lines denote patient RPM data, and dashed lines denote the response to patients’ record data via the pulse oximeter, which transmits to the Plan-it Med (PIM) App ® through Bluetooth technology. This data is read by a nurse or physician at Play-it Health, who summarizes weekly trends and reports findings to Three Village Allergy and Asthma, PLLC. RPM: remote patient monitoring.
Patients were instructed upon study enrollment to use the pulse oximeters once daily and when they suffered from respiratory symptoms. The study utilized Food and Drug Administration (FDA)-approved, high-fidelity, bluetooth-enabled iHealth pulse oximeters that interfaced through Play-it Health’s software PIM® with smartphones and tablets. Nurses and physicians reviewed clinical data daily 5 days per week (approximately 1 min per patient daily) and summarized monthly trends. We retrospectively examined the efficacy of the technology, using anonymously coded, de-identified data from a total of 102 patients who were enrolled in RPM at Three Village Allergy and Asthma, PLLC, South Setauket, New York, USA.
Platform Characteristics
The PiM platform interfaces with over 130 digital devices and displays data for patients and clinicians in real time on mobile or desktop devices. It supports five different methods of communication with patients. The platform has reminders and encourages verbiage for more meaningful engagement. The platform also has additional functionality such as education, survey administration, and rewards administration. Rewards were not used in this protocol.
Outcome Measurements
The following measurements were taken at the start of the study and at the end of 6 months.
Primary Outcome Measures
The primary outcomes for the extended follow-up were the number of physiological abnormalities, measures of compliance, out-of-pocket patient costs, insurance coverage, comorbidities, age, gender, and home zip codes. Physiological abnormalities were defined as a heart rate less than 60 and greater than 120 or pulse oximetry oxygen saturation less than 90% at rest. The responses between the on-call nurse and patients who decided to discontinue the service were compared with their most recent ACT responses collected before enrolling in RPM. The asthma control test (ACT) survey is a valid and reliable test whose scores reflect self-reported respiratory symptoms within the last 4 weeks. The average change in reported responses of these patients was compared with those of 24 patients who received at least 6 months of standard in-office clinical care who reported for an office visit during the study time frame.
Secondary Outcomes Measures
The secondary outcomes included the number of interventions made by the on-call nurse, which resulted in a change to the clinical plan of action. This may have included scheduling an appointment with an allergist/pulmonologist/cardiologist/primary care provider or visiting the Emergency Room as needed.
Statistical Analysis
Descriptive analyses were performed to characterize the sample and the study outcomes using the central tendency, confidence interval, and percentage measures. The level of significance was set at P < 0.05 for all tests. We used nonparametric rank tests to evaluate the effect of the time (before and after the intervention), and the normality of the distributions was verified using Wilcoxon signed-rank test for paired (dependent groups, before vs. after) and Wilcoxon rank-sum (Mann–Whitney) test for unpaired (independent groups, RPM vs. control). All models were adjusted for patient demographics and comorbidities. STATA software was used for all analyses.
Ethics
The study was submitted to the ethics committee of the Zucker School of Medicine at Hofstra/Northwell and was exempted from full review as only de-identified retrospective examination of an existing database administrative data was evaluated.
Results
Platform Engagement
A total of 102 patients were enrolled in the RPM study during the 14-month study (between February 3, 2020 and April 30, 2021). 66% of the patients were enrolled in February through April; 26% enrolled in May through July, and the remaining patients enrolled over the next 6 months. All patients were included in the full data analysis. The initial characteristics of the 102 patients are shown in Table 1.
| Active | Inactive | |
| Weight (SD) | 181.1 (47.1) | 172.9 (47.4) |
| Sample size (n) | 46 | 47 |
| P = 0.41 | ||
| BMI (SD) | 28.0 (7.5) | 28.0 (6.1) |
| Sample size (n) | 45 | 46 |
| P = 0.99 | ||
Detectable physiological abnormalities were noted among 19 patients (18.6%). Twenty-one RPM patients completed an ACTTM Questionnaire (29) (Table 2) during an office visit after being enrolled in RPM for at least 6 months during the study period. 82% of patients enrolled during months 1–6; 61% of patients uploaded data once every 24 h for 3 months after enrollment; 41.8% of patients utilized the system every 48 h for 6 months after enrolment. At 205 days after beginning the study, 50% of patients continued to report data to the RPM platform. At 365 and 700 days after study enrollment, 37.4 and 22.24%, respectively, of patients were still using the service (Fig. 2). Documentation of the messages between the on-call nurse and patients who decided to discontinue using RPM was reviewed, and the self-reported reasons provided by 35 of 54 patients (64.8%) were recorded and analyzed.
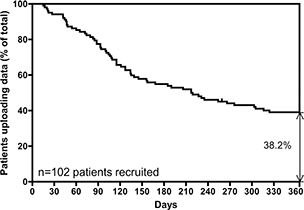
Fig. 2. Long-term RPM adherence by a group of 102 patients. One year after initiating pulse oximetry at home 38% of the patients were still reporting oxygenation and heart rate data through the app. RPM: remote patient monitoring.
Primary Outcomes
The RPM detected clinical findings that would not have been otherwise detected in 19 patients as these patients were asymptomatic. Twelve of 19 patients had abnormal heart rates, five patients had low oxygen saturations, and one patient had abnormal readings for both parameters. Twelve of 19 patients had a previous history of cardiovascular disease. Among these 12, eight patients had a history of hypertension, six patients had a history of structural heart disease (1 with aortic valve stenosis), two had atrial fibrillation, one had cardiomyopathy, one had mitral regurgitation and heart failure, and one had aortic aneurysms and coronary vessel stenosis.
Sixteen patients warranted consultation with a nonallergy/pulmonary specialist such as a cardiologist, and two patients were seen in the Emergency Room. After consulting with specialists, an additional two patients were newly diagnosed with heart disease, one with Coxsackie myocarditis, and another with bivalvular heart disease. Eight of 19 patients (42.1%) the RPM findings led to significant, actionable changes in the prescription regimens.
The ACT results of the asthmatic patients were collected in-office and were retrospectively assessed. Patients who used RPM for at least 6 months were compared with patients who had received at least 6 months of standard care for asthma during the same time period. On average, the RPM patients reported a greater decrease in instances of shortness of breath, as well as a significant decrease in their use of rescue inhalers and nebulizer medication (P = 0.005), and a nonsignificant increase in overall asthma control compared with the control group (P = 0.1) (Table 3).
In a subset analysis, 54 patients enrolled in RPM were longitudinally examined for 9 weeks, starting February 4, 2020, and through April 7, 2020. The average patient compliance with RPM tended to increase, with patients logging in daily for 78.9% of 2,350 patient days, weekly for 96.1% of 335 patient weeks, and monthly for 99.1% of 110 patient months (Fig. 3). The patients remained enrolled in the program for an average of almost nine months (272 days, n = 89). This included all nonpediatric patients who contributed at least 1 weeks’ worth of monitoring data.
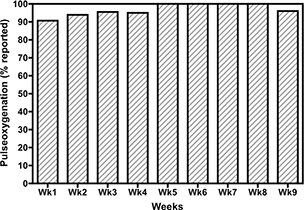
Fig. 3. Short-term adherence of 54 patients onboarded before April 7, 2020, with at least 1 week of reporting data, ranging from 9 to 64 days, plotted as the percentage of patients logging pulse oximetry data at least once per week.
Among the 43 patients enrolled and active in February 2021 who had been on the RPM platform for an average of 11 months, an average of 66.7 ± 6.0% reported a pulse oximetry values every 24 h. 82.1 ± 4.8% reported a value in any 48-h period (Fig. 4 and Table 4), demonstrates high levels of continuing engagement. Ten patients who either enrolled in RPM due to contracting COVID-19 or who contracted COVID-19 while enrolled in the program used the service for an average of 181 days, with two patients still active. Patients who contracted COVID-19 utilized the service 153 days, on average, before discontinuation, (n = 8). The compliance rates for RPM were assessed for active patients during February 2021 using the following criterion: a patient was considered ‘compliant’ if data were transmitted for 16 or more days in the month (55%). During February 2021, patients reported their vital signs on average 15 days out of the month (53.26%). 27 of 47 patients (57%) were considered compliant.
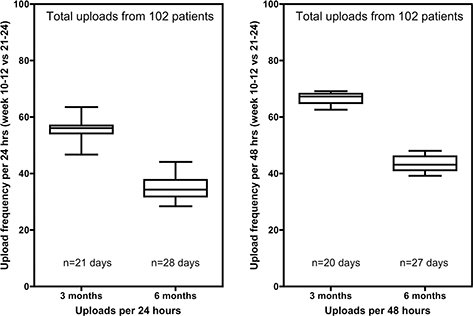
Fig. 4. Adherence to RPM reporting of pulse-oximetry data by 102 patients at 3 or 6 months after initiating the RPM reporting. The plots show the percentage of patients reporting in any given 24-h period (left plot) or 48-hour period (right plot). RPM: remote patient monitoring.
Qualitative Analysis of Patient Correspondence
Out of a total of 102 patients enrolled in RPM, 48 (47.1%) chose to continue to actively use the program (active patients are defined as regularly uploading their data). We chose to evaluate the data with a more stringent criteria than applied in other analyses, because of the clinical implications. 54 (52.9%) patients chose to discontinue or were removed from the program within 14 months (inactive patients). We examined correspondence (including notes on the monthly phone calls) between the on-call nurse and now inactive patients and noted that the self-reported reasons for discontinuing the program were determined in 36 of 54 patients (66.7%). The correspondence was reviewed and discussed by four of the authors (CM, KG, JD, AS) and key themes were identified. The three prominent reasons reported by patients as to why they discontinued the RPM program were 1) alleviation of symptoms for which the program was implemented (15 of 36 patients, 41.7%); 2) out-of-pocket costs borne by the patient (14 of 36 patients, 38.9%), and 3) difficulty using the program (6 of 36 patients, 16.7%). One patient (2.8%) was removed from the program after they were reported as deceased.
Active patients were, on average, older than inactive patients, with an average age of 59 years old (median age 62) versus an average age of 49 years old (median age 51). Women comprised most of both active and inactive patients, comprising 64.6% of active and 72.2% of inactive patients. Active patients tended to suffer from comorbid chronic health conditions more often than inactive patients, such as heart disease, diabetes, and thyroid disorders. Inactive patients tended to suffer from acute conditions more frequently, such as COVID-19 (Table 5). On average, the active patients’ insurance covered a higher percentage of the cost for RPM, covering 98.1% versus 92.5% for inactive patients. This difference was reflected in the average out-of-pocket costs to patients. Inactive patients tended to pay over three times more for RPM care than active patients, averaging $24.26 per month versus $7.19. No significant difference was found between the residential travel distance of the user from the practice and their status as an active versus inactive patient, with 2 of 48 (4.17%) active patients and 2 of 54 inactive patients (3.70%), living outside the county where the practice operates.
ACT Question Analysis
The results of the ACT (29) showed no difference in the response to questions 1–3 for the different groups. Both the control and RPM groups gave similar responses to ACT question 4 at the beginning of the study, but at the end of the study, and after the PIM intervention, the results showed much improvement in the RPM group. This represents a good example of the longitudinal study that was started with two similar groups (control and intervention with similar response) but at the end of the study, the intervention group showed statistically significant improvement based on question 4 (P = 0.005) with some trend in question 5 (P = 0.11). The issues addressed in Questions 4 and 5 have unique resonance in assessing asthma severity (Q4: during the past 4 weeks, how often have you used your rescue inhaler or nebulizer medication? and Q5: How would you rate your asthma control during the past 4 weeks?). These inferences are supported by the findings of the original ACT validation study that showed a strong and independent performance of these items (Q4 and Q5) in discriminating between patients who do and do not have controlled asthma, lending support to the suggestion that the functional impact (via ACT) can be routinely assessed when evaluating patients (30). This result suggests that RPM had a positive impact on reducing asthma severity.
Discussion
This is the largest study of remote patient telemonitoring of asthmatic patients at home. This study demonstrates that a noninvasive multiparameter telemonitoring system implemented in patients with asthma showed significant benefits. The RPM intervention attenuated the risks of asthma exacerbations leading to changes in clinical decision making. Notably, this RPM protocol was deployed in individuals with no to little prior experience or knowledge of RPM. The sole requirement for enrollment was access to a smartphone or tablet and English language comprehension.
The 3- and 6-month high engagement/adherence rates demonstrate the potential for this methodology in improving chronic illness care during a pandemic. The three key months of the study overlapped with the worst first wave of the US pandemic crisis during which access to in-person clinical care was significantly decreased to unavailable for nonemergent situations. A significant proportion of these patients chose to continue these services up to 2 years after enrollment. Though patients were initially asked to use the platform daily when access to in-person care was decreased, they were only expected to use the platform every other day after their clinical condition stabilized. Patients whose respiratory symptomatology resolved were not expected to continue to use the platform but rather to have the platform as a tool for future use as needed.
The main primary study endpoint benefits were seen in patients who actively used RPM for at least 6 months and who reported increased asthma control when compared with controls. This was demonstrated in the average change in reported scores for 3 of 5 ACT questions, as well as the average change in the overall scores (Tables 2 and 3). Approximately 19% of patients studied received some form of medical intervention earlier than when using routine non-RPM care. These interventions led to earlier diagnoses of medical conditions in two of the 19 patients (11%) and to changes in prescribed medications in 8 of 19 patients (42%). For those patients, the early detection provided by the RPM technology may have attenuated the risk of going to the emergency room secondary to worsening symptoms. For two of 19 patients (11%), these interventions led to an earlier arrival to the emergency room, possibly increasing their chances of survival. The results from these key study periods suggest that the reduction in morbidity in the RPM group was not completely attenuated after 6 months and supports the generalizability and sustainability of the results.
In addition, these results offer insights for future evaluation of the technology. Two common trends were seen in the population studied: 1) patients suffering from acute conditions used the technology to send data to a medical professional for monitoring until their symptoms subsided and 2) patients suffering from non-pulmonary chronic comorbid conditions alongside their chronic respiratory conditions used the technology as a long-term ‘safety net’, more often sending data when symptoms arose. The distinction between these two types of users is important, the former being considered a ‘short-term’ user and the latter considered a ‘long term’ user should be addressed in further evaluation of RPM effectiveness. Similarly, the dropout of short-term users over time should not necessarily be seen as evidence for a lack of effectiveness of patient adherence in the evaluation of RPM. A very relevant but still unanswered question concerns what the ideal duration of noninvasive RPM is when used in a real-world setting of standard care asthmatic patients. This study, therefore, suggests that there are two subgroups that could clearly benefit from RPM and for which this system would be very effective: 1) those needing acute management of medical conditions, especially when usual clinical contact may be reduced and 2) those with chronic conditions that would benefit from a longer-term chronic care management. The long-term adherence rates in the subgroup that desired long-term management is some of the highest compliance rates ever reported.
Home Monitoring Implications
There are a wide variety of home monitoring and surveillance strategies, but there are very few studies that report on the use of ACT and pulse oximetry devices (31). The findings of this study reflect known factors that increase the adherence to RPM, emphasizing the importance of the system’s affordability, ease of use, and limited interruption of patients’ daily lives (32, 33). As a result, a number of changes in RPM policy are in order to help increase the implementation efficacy of RPM technology.
It is clear that patient selection is critical to increasing patient adherence and decreasing dropout rates with regard to RPM. Before a patient is enrolled in the program, patients should be informed about out-of-pocket insurance monthly charges. This will assist patients in making informed decisions and managing their expectations. Development and identification of more cost-effective devices will help decrease patient out-of-pocket costs, leading to decreased inactivity due to cost-related concerns. Improvements in the onboarding process could potentially improve patient adherence as well as health outcomes. At the start of the onboarding process, the intended purpose of starting the program should be identified, and the patient should be designated as either a short-term user or long-term user, as described above. Short-term users could be encouraged to restart using the program at any time if they start developing any symptoms, and long-term users could be encouraged to incorporate RPM into their daily routine alongside acute events. During the onboarding process, patients should be trained in both the transmission of data and the use of the messaging system to mitigate dropouts due to frustration. The implementation of these policies could significantly improve the efficacy of RPM for patients with acute or chronic dyspnea.
Finally, the importance of access to a mobile platform during periods of decreased access to care such as during the COVID-19 pandemic cannot be overemphasized (34). Patients frequently reported comfort in securely knowing they were able to maintain remote contact with their care providers during the pandemic when their access to in-person visits was highly constrained. Future studies are needed to better quantify this effect (35).
There are several limitations of this study. First, although the sample size in the current study is small, this is a preliminary investigation regarding an alternative method for home supervision including medication adherence. It could improve access to this type of program for asthmatic patients with a risk profile that is greater than has been previously reported. Second, its retrospective design may impact what can be reliably inferred, but the robust findings and high compliance and RPM loyalty rates are noteworthy. Third, we made significant efforts to address any confounding factors that might shed light on the RPM vs the control group different ACT data. While there could be other confounding reasons for the measured ACT differences, we did not see any that would explain our findings in the RPM group as compared with the control group. Fourth, the study was conducted during the COVID-19 pandemic, which potentially could affect patient RPM compliance, as well as patients’ decisions to continue using the program.
Additionally, patient home self-isolation could have led to increased adherence and continuation with the program, due to increased social interaction they might not have otherwise had (36). However, the self-isolation caused by the pandemic may also have contributed to the frequency of patients choosing to discontinue the program due to frustrations with the technology, as any technical problems that arose had to be solved through phone or remote video calls rather than in-person communication. Patients who were onboarded due to contracting COVID-19 typically discontinued after recovery, as they felt that they no longer required home monitoring.
The results of this study can help with the design of home-based RPM programs adapted to clinical situations with moderate-risk asthmatic and COPD patients and also in the design of new technologies adapted to improve medication and inhaler compliance and self-care.
Conclusions
A home-based asthmatic program with a hybrid surveillance program demonstrates the positive effects of an RPM intervention on morbidity related to acute and chronic pulmonary dyspnea, early detection of physiological abnormalities, and early medical interventions. Long-term compliance was excellent but limited by resolution of disease, financial constraints, and technical issues. High adherence levels were associated with older age, comorbidities, and having Medicare Insurance. Our results are thus exploratory and further long-term research is needed to determine what the ideal RPM technology and duration of noninvasive RPM should be when used in real-world settings. Additionally, profiling of the asthmatic patient population most likely to benefit from the noninvasive RPM intervention is needed. Conducting research on the costs and RPM performance is still at an early stage, and further studies are needed to determine whether this study’s results are consistent across other regions and healthcare systems.
Contributors
Study design and patient enrollment were performed by Dr. Szema and Dr. Gandy. The initial data collection and analysis and preparation of the manuscript were performed by Dr. Marsh under the supervision of Dr. Szema, Dr. Gandy, and Dr. Domen. Extensive revision of the manuscript was performed by Dr. Barach. Additional data collection and analysis were performed by Mr. Chen. Statistical analysis was performed by Dr. Hamidi
Acknowledgments
None
References
- Gates B. Shattuck lecture innovation for pandemics. Presented at: Shattuck Lecture Innovation for Pandemics, 27 April 2018. Available from: https://www.gatesfoundation.org/Ideas/Speeches/2018/04/Shattuck-Lecture-Innovation-for-Pandemics [cited 2 February 2022].
- Fineberg H. Pandemic preparedness and response – lessons from the H1N1 influenza of 2009. N Engl J Med 2014; 370(14): 1335–42. doi: 10.1056/NEJMra1208802
- Whitelaw S, Mamas AM, Topol E, Van Spall HGC. Applications of digital technology in COVID-19 pandemic planning and response. Lancet Digit Health 2020; 2(8): E435–40. doi: 10.1016/S2589-7500(20)30142-4
- Pevnick JM, Fuller G, Duncan R, Spiegel BMR. A large-scale initiative inviting patients to share personal fitness tracker data with their providers: initial results. PLoS One 2016; 11: e0165908. doi: 10.1371/journal.pone.0165908
- Andreu-Perez J, Leff DR, Ip HM, Yang GZ. From wearable sensors to smart implants – toward pervasive and personalized healthcare. IEEE Trans Biomed Eng 2015; 62: 2750–62. doi: 10.1109/TBME.2015.2422751
- Ajami S, Teimouri F. Features and application of wearable biosensors in medical care. J Res Med Sci 2015; 20: 1208–15. doi: 10.4103/1735-1995.172991
- Wong AH, Ahmed RA, Ray JM, Khan H, Hughes PG, McCoy CE, et al. Supporting the Quadruple aim using simulation and human factors during COVID-19 care. Am J Med Qual Off J Am Coll Med Qual 2021; 36(2): 73–83. doi: 10.1097/01.JMQ.0000735432.16289.d2
- Behar JA, Liu C, Kotzen K, Tsutus K, Corino VDA, Singh J et al. Remote health diagnosis and monitoring in the time of COVID-19. Physiol Meas 2020; 41(10): 10TR01. doi: 10.1088/1361-6579/abba0a
- Dunn P, Hazzard E. Technology approaches to digital health literacy. Int J Cardiol 2019; 293: 294–6. doi: 10.1016/j.ijcard.2019.06.039
- Su D, Michaud TL, Estabrooks P, Schwab RJ, Leslie A, Eiland LA, et al. Diabetes management through remote patient monitoring: the importance of patient activation and engagement with the technology. Telemed E-Health 2018; 25(10): 952–9. doi: 10.1089/tmj.2018.0205
- Lee PA, Greenfield G, Pappas Y. The impact of telehealth remote patient monitoring on glycemic control in type 2 diabetes: a systematic review and meta-analysis of systematic reviews of randomised controlled trials. BMC Health Serv Res 2018; 18(1): 495. doi: 10.1186/s12913-018-3274-8
- Huang Z, Tao H, Meng Q, Jing L. Management of endocrine disease. Effects of telecare intervention on glycemic control in type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Eur J Endocrinol 2015; 172(3): R93–101. doi: 10.1530/EJE-14-0441
- Medical Advisory Secretariat. Home telemonitoring for type 2 diabetes: an evidence-based analysis. Ont Health Technol Assess Ser 2009; 9(24): 1–38.
- Zhai YK, Zhu WJ, Cai YL, Sun DX, Zhao J. Clinical- and cost-effectiveness of telemedicine in type 2 diabetes mellitus: a systematic review and meta-analysis. Medicine (Baltimore) 2014; 93(28): e312. doi: 10.1097/MD.0000000000000312
- Saffari M, Ghanizadeh G, Koenig HG. Health education via mobile text messaging for glycemic control in adults with type 2 diabetes: a systematic review and meta-analysis. Prim Care Diabetes 2014; 8(4): 275–85. doi: 10.1016/j.pcd.2014.03.004
- Hanlon P, Daines L, Campbell C, McKinstry B, Weller D, Pinnock H. Telehealth interventions to support self-management of long-term conditions: a systematic metareview of diabetes, heart failure, asthma, chronic obstructive pulmonary disease, and cancer. J Med Internet Res 2017; 19(5): e172. doi: 10.2196/jmir.6688
- Hale TM, Jethwani K, Kandola MS, Saldana F, Kvedar JC. A remote medication monitoring system for chronic heart failure patients to reduce readmissions: a two-arm randomized pilot study. J Med Internet Res 2016; 18(4): e91. doi: 10.2196/jmir.5256
- Abraham WT, Adamson PB, Bourge RC, Bourge RC, Aaron MF, MD, Costanzo MR, et al. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: a randomised controlled trial. Lancet 2011; 377(9766): 658–66. doi: 10.1016/S0140-6736(11)60101-3
- Alotaibi S, Hernandez-Montfort J, Ali OE, El-Chilali K, Perez BA. Remote monitoring of implantable cardiac devices in heart failure patients: a systematic review and meta-analysis of randomized controlled trials. Heart Fail Rev 2020; 25(3): 469–79. doi: 10.1007/s10741-020-09923-1
- Givertz MM, Stevenson LW, Costanzo MR, Bourge RC, Bauman JG, Ginn G, et al. Pulmonary artery pressure-guided management of patients with heart failure and reduced ejection fraction. J Am Coll Cardiol 2017; 70(15): 1875–86. doi: 10.1016/j.jacc.2017.08.010
- Cruz J, Brooks D, Marques A. Home telemonitoring effectiveness in COPD: a systematic review. Int J Clin Pract 2014; 68(3): 369–78. doi: 10.1111/ijcp.12345
- McLean S, Nurmatov U, Liu JL, Pagliari C, Car J, Sheikh A. Telehealthcare for chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2011; (7): CD007718. doi: 10.1002/14651858.CD007718.pub2
- Kamei T, Yamamoto Y, Kajii F, Nakayama Y, Kawakami C. Systematic review and meta-analysis of studies involving telehome monitoring-based telenursing for patients with chronic obstructive pulmonary disease. Jpn J Nurs Sci 2013; 10(2): 180–92. doi: 10.1111/j.1742-7924.2012.00228.x
- Murphy LA, Harrington P, Taylor SJ, Yuki Nakayama, Chiharu Kawakami, Pinnock H, et al. Clinical-effectiveness of self-management interventions in chronic obstructive pulmonary disease: an overview of reviews. Chron Respir Dis 2017; 14(3): 276–88. doi: 10.1177/1479972316687208
- Shah SA, Velardo C, Farmer A, Tarassenko L. Exacerbations in chronic obstructive pulmonary disease: identification and prediction using a digital health system. J Med Internet Res 2017; 19(3): e69. doi: 10.2196/jmir.7207
- Cooper CB, Sirichana W, Arnold MT, Teljeur C, Smith SM, Pinnock H, et al. Remote patient monitoring for the detection of COPD exacerbations. Int J Chron Obstruct Pulmon Dis 2020; 15: 2005–13. doi: 10.2147/COPD.S256907
- Mosnaim GS, Stempel DA, Gonzalez C Kaye L, Shalowitz M, Szefler S, et al. The impact of patient self-monitoring via electronic medication monitor and mobile app plus remote clinician feedback on adherence to inhaled corticosteroids: a randomized controlled trial. J Allergy Clin Immunol Pract 2021; 9(4): 1586–94. doi: 10.1016/j.jaip.2020.10.064
- Kew KM, Cates CJ. Home telemonitoring and remote feedback between clinic visits for asthma. Cochrane Database Syst Rev 2016; (8): CD011714. doi: 10.1002/14651858.CD011714.pub2
- Schatz M, Sorkness CA, Li JT, Marcus P, Murray JJ, Nathan RA, et al. Asthma control test: reliability, validity, and responsiveness in patients not previously followed by asthma specialists. J Allergy Clin Immunol 2006; 117(3): 8. doi: 10.1016/j.jaci.2006.01.011
- Nathan RA, Sorkness CA, Kosinski M, Schatz M, Li JT, Marcus P, et al. Development of the asthma control test: a survey for assessing asthma control. J Allergy Clin Immunol 2004; 113(1): 59–65. doi: 10.1016/j.jaci.2003.09.008
- Piotrowicz E, Baranowski R, Bilinska M, Stepnowska M, Piotrowska M, Wójcik A, et al. A new model of home-based telemonitored cardiac rehabilitation in patients with heart failure: effectiveness, quality of life, and adherence. Eur J Heart Fail 2010; 12(2): 164–71. doi: 10.1093/eurjhf/hfp181
- Majumder S, Mondal T, Deen MJ. Wearable sensors for remote health monitoring. Sensor 2017; 17: 130. doi: 10.3390/s17010130
- Institute of Medicine (US) Committee on Quality of Health Care in America. Crossing the quality chasm: a new health system for the 21st century. National Academies Press (US); 2001. Available from: http://www.ncbi.nlm.nih.gov/books/NBK222274/ [cited 1 July 2021].
- Romani G, Dal Mas F, Massaro M, Cobianchi L, Modenese M, Barcellini A et al. Population health strategies to support hospital and intensive care unit resiliency during the COVID-19 pandemic: the Italian experience. Popul Health Manag 2021; 24(2): 174–81. doi: 10.10931089/pop.2020.0255
- Piras EM, Miele F. On digital intimacy: redefining provider-patient relationships in remote monitoring. Sociol Health Illn 2019; 41(Suppl 1):116–31. doi: 10.10931111/1467-9566.12947
- Parretti C, Tartaglia R, La Regina M, Venneri F, Sbrana G, Mandò M., Improved FMEA methods for proactive health care risk assessment of the effectiveness and efficiency of COVID-19 remote patient telemonitoring. Am J Med Qual 2022; 37(6): 535–544. doi: 10.10931097/JMQ.0000000000000089
Copyright Ownership: This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0