ORIGINAL RESEARCH
Internet of Things Technologies and Applications in the Management of Diabetes Complications: A Systematic Review
Olapeju Ajibade, PhD student, MSc (Adult Health Nursing), BNSc, (Nursing)1  ; Oluwaseyi Akpor, PhD, MSc, BNSc2
; Oluwaseyi Akpor, PhD, MSc, BNSc2  ; Sunday Afolalu, PhD, MSc3
; Sunday Afolalu, PhD, MSc3  ; Gloria Oluwakorede, Alao, BNSc, RN, RM, RPHN4
; Gloria Oluwakorede, Alao, BNSc, RN, RM, RPHN4  ; Bose Ogunlowo, BNSc5
; Bose Ogunlowo, BNSc5  ; Oluwatosin Ogunmuyiwa, MPH, BNSc (Nursing)6
; Oluwatosin Ogunmuyiwa, MPH, BNSc (Nursing)6  and Oluwadamilare Akingbade, PhD, MSc, CGNC, RN7
and Oluwadamilare Akingbade, PhD, MSc, CGNC, RN7 
1PhD Student, Faculty of Nursing, Afe Babalola University, Ado-Ekiti, Nigeria; 2Professor, Department of Nursing, Saint John Regional Hospital, Saint John New Brunswick, Canada; 3Professor, Faculty of Engineering, Afe Babalola University, Ado-Ekiti, Nigeria; 4Nursing Officer, Wilson and Paulina Memorial Hospital Onuaku, Abia State, Nigeria; 5Lecturer, Department of Nursing, Obafemi Awolowo University, Osun, Nigeria; 6Lecturer I, Faculty of Nursing, University of Medical Sciences, Ondo, Ondo State, Nigeria; 7Postdoctoral Fellow/Lecturer, Faculty of Nursing, University of Alberta, Edmonton, Alberta, Canada
Keywords: Chronic disease management, diabetes monitoring, Internet of Things, IoT devices, diabetes complications
Abstract
Background: The increasing prevalence, complications, and mortality associated with diabetes worldwide create an urgent need for innovative approaches to chronic disease management. In response, the Internet of Things (IoT) has been increasingly integrated into healthcare systems to enhance monitoring, early detection, and timely intervention for diabetes-related complications. These IoT-enabled interventions use interconnected devices such as continuous glucose monitors, wearable sensors, and mobile health applications to collect real-time patient data, facilitate remote monitoring, and support personalized treatment plans. This systematic review critically evaluates the effectiveness of such IoT-based solutions in improving the quality of life and management of diabetes.
Methods: A comprehensive search of Web of Science, CINAHL, Scopus, PubMed, Embase, and Medline was conducted. Data were extracted on intervention design, IoT components, outcomes, and theoretical foundations.
Results: The search results included 2,436 articles, from which 17 randomized controlled trials that were published between 2010 and 2024. Among the included studies, 82.4% employed true randomization, while 5.9% reported blinding of outcome assessors. Mobile applications were the most frequently used IoT tools, often integrated with Bluetooth-enabled glucometers, pedometers, or web-based platforms. Significant improvements in hemoglobin A1c were observed in most interventions, particularly those with personalized feedback, real-time data transmission, and healthcare provider involvement. A few studies also reported improvements in self-care, diet adherence, and distress reduction. However, theoretical underpinnings, standardization, and long-term outcome reporting were largely absent.
Conclusion: The IoT technologies offer promising support for glycemic control and complication management in diabetes care. Interventions that combine education, behavioral support, and bidirectional feedback demonstrate the greatest impact. Future research should focus on standardized, theory-driven designs and long-term evaluations to optimize patient outcomes and ensure equitable access.
Plain Language Summary
This study reviewed 17 research papers published between 2010 and 2024 that tested how smart devices like mobile apps, Bluetooth glucometers, and telehealth platforms can help people with diabetes detect and manage health complications. Most of these tools helped lower blood sugar levels, especially those that gave personalized messages and connected patients with healthcare professionals. Tools that used real-time feedback, such as those linked with wearable sensors and monitoring systems, helped patients stay engaged with their treatment and make healthier lifestyle choices. While these technologies show great promise, future research should ensure they are easier to use, grounded in behavior-change science, and accessible to people everywhere.
Citation: Telehealth and Medicine Today © 2025, 10: 582
DOI: https://doi.org/10.30953/thmt.v10.582
Copyright: © 2025 The Authors. This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, and the use is non-commercial. See http://creativecommons.org/licenses/by-nc/4.0.
Submitted: April 27, 2025; Accepted: May 30, 2025; Published: September 24, 2025
Corresponding Author: Ajibade Olapeju, Email: ooajibade@unimed.edu.ng
Competing interests and funding: No relevant disclosures.
None.
Diabetes mellitus (DM) is a chronic condition, and continuous monitoring is the key to effective management.¹ It impacts multiple physiological systems that can lead to serious complications such as morbidity and healthcare costs, along with increased risk of mortality.²,³ Common complications include cardiovascular disease, kidney disease, retinopathy, neuropathy, and depression. The World Health Organization (WHO) categorizes these complications into microvascular and macrovascular disorders.4 Microvascular complications are often manifested by markers such as proteinuria, elevated serum creatinine, body weight changes, fasting blood glucose (FBG) levels, and blood pressure. Factors such as age, duration of diabetes, coexisting health conditions, and poor glycemic control also significantly influence the development and progression of these complications.5,6
Furthermore, in 2021, diabetes was the direct cause of 1.6 million deaths, and 47% of all deaths due to diabetes occurred before the age of 70 years.7 The International Diabetes Foundation estimates that 589 million adults worldwide are living with diabetes, and this is projected to increase to 853 million by 2050.8
The introduction of Internet of Things (IoT) technology has transformed various industries globally, including the healthcare sector. Patients with DM will benefit greatly from the rapidly evolving technological growth of IoT.9 The IoT comprises the integration of different kinds of physical findings, types of equipment, and sensors via the Internet to enable them to communicate with each other and share data.10
Today, IoT technologies are used in the healthcare sector to improve patient care and outcomes, offering new opportunities for remote monitoring, personalized treatment plans, and efficient healthcare delivery.11 The ability of IoT mobile applications to facilitate real-time data collection, remote monitoring, and personalized feedback makes them suitable for managing disease conditions like diabetes.12
Mobile applications that provide solutions to several challenges faced by patients with diabetes include Io T-enabled glucometers and continuous glucose monitors that provide real-time tracking of blood glucose levels and can alert patients and healthcare providers to abnormal readings. Medication management apps remind patients to take their medications as well as track adherence. Fitness trackers and dietary monitoring apps help patients maintain a healthy lifestyle by providing personalized recommendations and feedback.13 Wearable IoT devices, like glucose monitoring systems and insulin pumps, offer the potential to improve patient care, prevent disease progression, and reduce costs—ultimately improving life quality for patients. These devices are becoming more sophisticated at collecting and analyzing data in real time. Technical challenges and significant barriers faced with the use of IoT technologies include interoperability between devices, data security and privacy concerns, and the need for reliable wireless connectivity.14
In this study, the authors systematically review the use of IoT technologies for the management of diabetes and its associated complications. The objectives are (1) identify and categorize the various IoT devices and technologies currently used in diabetes complication management; (2) analyze the design features and components of these IoT tools; (3) evaluate their impact on key health indicators such as weight, body mass index (BMI), blood glucose concentrations, waist circumference, and cholesterol; and (4) assess the overall effectiveness of IoT technologies in managing diabetes-related complications.
Methods
This study used the Preferred Reporting Items for Systematic Study and Meta-Analysis (PRISMA) protocol to examine how IoT affects early diagnosis and management of complications from 2010 to 2024.15 Protocol number 628975 was registered with the International Prospective Register of Systematic Reviews (aka: PROSPERO). Key data from each study included authors’ names, publication details, country, objectives, methodology, outcomes, and conclusions, summarized narratively. The IoT effects studies published in English in peer-reviewed journals were included, with comprehensive searches using Web of Science, CINAHL, Scopus, PubMed, Embase, Medline, and reference scanning for additional source (Figure 1).
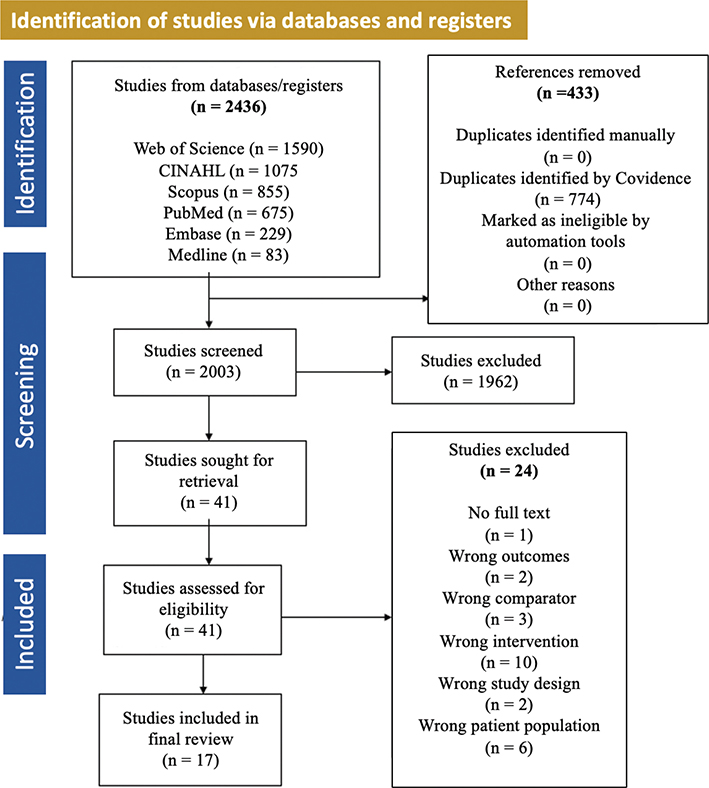
Fig. 1. PRISMA flow diagram showing the study selection process. PRISMA: Preferred Reporting Items for Systematic Study and Meta-Analysis.
The study examines how IoT affects diabetes control. Models, devices, hemoglobin A1c (HbA1c), BMI, weight, blood pressure, FBG, diabetes mellitus, complications, and people living with diabetes are search phrases. Mesh terms include early detection, monitoring, treatment, and complications.
The search was confined to health indicators, language (English), publication type, and dates from 2010 to 2024 (Table 1).
Results
Characteristics of Included Studies
A total of 17 studies were analyzed. Of these, 14 (82.4%) employed true randomization for group allocation.16,18–20,22–32 However, only one study (5.9%) clearly reported blinding of outcome assessors,16 and four (23.5%) were unclear regarding the blinding status of those delivering the interventions.18,22,27,30 The majority of interventions (n = 14) focused on mobile applications, web-based programs, and telehealth services16–18,20,22–24,26–32 These included educational text messages, glucose-tracking applications, and telemonitoring devices. Several studies also used cloud-based platforms and text-messaging systems.19,24,31
The HbA1c, a widely accepted marker of glycemic control, was the primary outcome across all 17 studies.16–32 In terms of delivery modality, 14 interventions were app based,16,18,20,22–24,26–32 while others incorporated web, cloud, or telemonitoring systems.19,23,30 Seven studies had a 6-month duration,16,18–22 four lasted 12 months,23–26 and the remaining varied between 1 and 11 months.27–32 As summarized in the Appendix, IoT interventions comprised mobile apps, web-based platforms, and integrated health systems. These were delivered through a combination of educational materials, Bluetooth-enabled glucose tracking devices, telemonitoring equipment, and comprehensive health platforms.16,18–20,22–24,28–30
The frequency of intervention delivery ranged from daily (e.g. Dulce Digital,16 DiaNote26), to weekly19,27 or multiple times per week.18,20,22 Most studies featured a two-arm design; however, three studies employed two experimental arms.16,20,28 Only one study explicitly reported grounding its intervention in theoretical models namely, the Knowledge-Attitude-Practice (KAP) model and the Health Belief Model.22
IoT Technologies Employed
The dominant IoT technologies used in the reviewed studies included mobile applications, web-based platforms, and connected healthcare systems.16–32 These tools were employed to deliver educational content, track blood glucose, facilitate remote monitoring, and support integrated care. Interventions such as Dulce Digital16 and DiaNote,26 which utilized text messaging and app-based interfaces, showed significant reductions in HbA1c. Studies incorporating Bluetooth-enabled devices23 and telemonitoring systems29 demonstrated improved glycemic control and enhanced patient engagement, contributing to better adherence to treatment regimens. Interventions that combined educational content with behavior modification strategies further led to improvements in diet, physical activity, and medication adherence.22
Outcomes of the Included Studies
The HbA1c was the primary outcome measure in all studies, assessed using various validated techniques.16–32 In nearly all studies, intervention groups showed greater reductions in HbA1c levels compared to control groups. Additional outcomes included changes in lipid profiles and BMI, as observed in several studies.16,18,23 A limited number of’ studies also assessed clinical complications such as severe hypoglycemia and diabetic ketoacidosis.17,19 Several studies incorporated patient-reported outcomes, including diabetes-related distress,27 dietary adherence,22 and self-management abilities.29
Effects of IoT Technologies on Diabetes Complications
The findings indicate that mobile apps, text messaging systems, and web-based platforms can effectively lower HbA1c levels.16–32 Notably, interventions with personalized feedback and physician involvement, such as those described by16,20 yielded greater clinical benefits. Bluetooth-enabled devices23 and telemonitoring tools29 contributed to improved glycemic control and greater adherence to treatment plans. Furthermore, combining educational interventions with behavior change strategies significantly enhanced adherence to diet, physical activity, and medication.22
Figure 2 summarizes the reductions in HbA1c achieved across various digital interventions. The most notable reduction (1.80%) was reported in a study involving adolescents using glucose tracking applications.17 Other significant reductions included 0.65%19 and 0.52%.23 Some studies noted improvements but did not provide specific numerical values.18,26 These are marked as ‘NR’ (not reported) in the figure. Collectively, the graph highlights the diverse efficacy of IoT-enabled interventions in improving glycemic control among patients with diabetes.
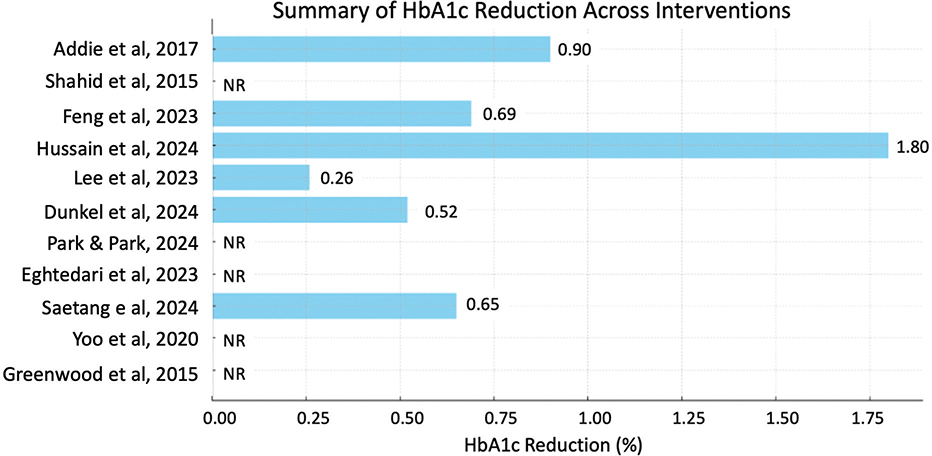
Fig. 2. Summary of HbA1c. HbA1c: hemoglobin A1c, NR: not reported.
Discussion
This systematic review correlated evidence on the use of IoT-enabled interventions in diabetes care, with a focus on early detection and management of complications. To our knowledge, this is the first comprehensive review dedicated to examining the effectiveness of IoT technologies in managing diabetes-related complications. The findings contribute important insights that can inform the design and implementation of more responsive, patient-centered IoT interventions for people living with diabetes.
The majority of interventions included in this review were built on mobile health applications such as DiaNote, iCareD, and Healthynote, which support education, behavioral prompts, and remote data tracking via smartphones and tablets.20,26,27 Many of these apps incorporated features such as blood glucose monitoring, dietary logging, and automated notifications that were often paired with Bluetooth-enabled glucometers, pedometers, and digital scales for continuous biometric monitoring.16,23 Web-based platforms and home telehealth systems also played a key role in enabling healthcare providers to monitor patients remotely, offer personalized feedback, and adjust treatment plans accordingly.29,30 One systematic review revealed that telehealth interventions have a positive impact on self-management among diabetic patients.31 Similarly, there are studies that support the effectiveness of mHealth interventions in enhancing remote monitoring for patients.32–34
The design of these technologies emphasized real-time data exchange and user-centered education. Interventions like Dulce Digital and DiaNote demonstrated how structured educational messages and behavior-modification reminders could improve glycemic outcomes.16,26 In some cases, multimedia content such as instructional videos and infographics was used to enhance health literacy and engagement.27 A recurring theme across the most effective interventions was the inclusion of personalized feedback and active healthcare provider involvement. For instance, bidirectional communication between patients and healthcare teams, as observed in iCareD, was associated with greater adherence and more favorable clinical outcomes.20
Most studies demonstrated significant reductions in HbA1c, reaffirming the utility of IoT technologies in glycemic control. Notable examples include a 0.69% HbA1c reduction through a WeChat-based intervention in China22 and a 1.8% reduction in Pakistani adolescents using an Urdu-language glucose tracking app.17 Additionally, integrating telemonitoring with automated feedback loops significantly improved glycemic outcomes.29 While there was some evidence of minor improvements in BMI and weight loss,28 these effects were not uniformly significant across all populations or time points.19 Still, many interventions enhanced dietary adherence, physical activity, and self-efficacy, reinforcing the behavioral potential of IoT applications in diabetes management.22,35 It is reported that although the IoT-based approach significantly promoted patients’ behavioral changes in exercise and diet, HbA1c levels were not reduced long term, as some patients in the intervention group did not use IoT despite adequate guidance.24
Importantly, several interventions supported the early detection and management of complications. Devices such as smart glucometers and pedometers encouraged active self-monitoring, helping patients identify trends in blood glucose and physical activity. Education-based platforms helped reduce diabetes distress and improve psychological outcomes.27 However, effectiveness varied across demographic subgroups and was often influenced by digital literacy, age, disease severity, and the complexity of the technological interface. In addition, one study also corroborated the findings of this study, as it showed that mHealth interventions significantly improve psychoeducational support among patients.36
Limitations
Despite the promise of these technologies, several limitations emerged. A significant proportion of studies did not ground their interventions in established behavioral theories, limiting understanding of the mechanisms behind their effectiveness and reducing the replicability of results. Only one study explicitly applied models like the Knowledge-Attitude-Practice framework and the Health Belief Model,22 highlighting a broader need for theoretically informed digital health design. Additionally, the short duration of many interventions and small sample sizes restricted the ability to assess long-term impacts and generalize findings across populations.37 There were also inconsistencies in outcomes: some trials employing advanced technologies failed to demonstrate significant changes in HbA1c,21,24 which raises questions about user engagement, intervention fidelity, and implementation barriers.
Further, heterogeneity in intervention components, such as the frequency of messaging, degree of healthcare provider involvement, and app features, makes cross-study comparisons difficult. Most studies did not systematically report engagement rates, user satisfaction, or adverse events, omitting key metrics for evaluating feasibility, acceptability, and safety. Moreover, while clinical markers like HbA1c were prioritized, few studies assessed psychosocial or economic outcomes such as quality of life, mental health, or cost-effectiveness, which are critical dimensions for holistic diabetes care.
Conclusion
In summary, IoT technologies hold transformative potential for diabetes management by enabling continuous monitoring, real-time feedback, and personalized care pathways. Tools such as smart glucometers, fitness wearables, and mobile apps have shown consistent benefits in glycemic control and in promoting healthier behaviors. However, issues such as lack of standardization, limited theoretical grounding, digital access disparities, and underreporting of engagement outcomes remain pressing challenges. To fully harness the potential of IoT in diabetes care, coordinated efforts involving healthcare providers, developers, researchers, and policymakers are essential. Emphasis should be placed on developing robust, theory-based interventions, establishing standard protocols for implementation, and evaluating both clinical and psychosocial outcomes over longer periods. With strategic investment and inclusive design, IoT-enabled care can substantially improve the quality of life for millions of individuals living with diabetes, particularly in low-resource and digitally underserved settings.
Contributors
Each author contributed the following aspects of the study: Dr. Ajibade: conceptualization, methodology, investigation, and writing original draft preparation; Dr. Akpor: conceptualization, review, and supervision; Dr. Afolalu: conceptualization, review, and supervision; Ms. Alao: conceptualization, methodology, writing original draft, and editing. Ms. Ogunlowo: methodology, writing original draft, and editing; Ms. Oluwatosin, Ogunmuyiwa: methodology, writing original draft, writing review, and editing; Dr. Akingbade: methodology, software, validation, formal analysis, review, and editing. All authors have read and agreed to the published version of the manuscript.
Data availability Statement (DAS), Data Sharing, Reproducibility, and Data Repositories
All data generated or analyzed during this systematic review are included in this published article and its supplementary materials. Additional materials such as the search strategy, PRISMA checklist, and data extraction forms are available upon reasonable request. The review methodology strictly followed PRISMA 2020 guidelines, and all steps are documented to ensure reproducibility.
Application of AI-Generated Text or Related Technology
The section summarizing HbA1c reductions across interventions was initially prepared with the aid of ChatGPT. All content was subsequently reviewed, edited, and approved by the authors. Artificial intelligence tools were not used independently to generate or analyze study results.
References
- Valsalan P, Hasan NU, Farooq U, Zghaibeh M, Baig I. IoT based expert system for diabetes diagnosis and insulin dosage calculation. Healthcare (Basel). 2023;11(1):12. https://doi.org/10.3390/healthcare11010012
- Ali, et al. A novel approach for best parameters selection and feature engineering to analyze and detect diabetes: machine learning insights. BioMed Res Int. 2023;8583210. https://doi.org/10.1155/2023/8583210
- Deepa R, Sivasamy A. Advancements in early detection of diabetes and diabetic retinopathy screening using artificial intelligence. AIP Adv. 2023;13(11). https://doi.org/10.1063/5.0172226
- Aikaeli F, Njim T, Gissing S, Moyo F, Alam U, Mfinanga SG, et al. Prevalence of microvascular and macrovascular complications of diabetes in newly diagnosed type 2 diabetes in low-and-middle-income countries: a systematic review and meta-analysis. PLoS Glob Public Health. 2022;2(6):e0000599. https://doi.org/10.1371/journal.pgph.0000599
- Gashaye SN, Sharew IA. Predictors of blood glucose change and microvascular complications of type 2 DM patients in FelegeHiwot and Debre Markos referral hospital, North West Ethiopia. BMC Endocr Disord. 2022;22(1):136. https://doi.org/10.1186/s12902-022-01047-x
- Seid MA, Akalu Y, Gela YY, Belsti Y, Diress M, Fekadu SA, et al. Microvascular complications and its predictors among type 2 DM patients at Dessie town hospitals, Ethiopia. Diabetol Metab Syndr. 2021;13(1):86. https://doi.org/10.1186/s13098-021-00704-w
- World Health Organization. Diabetes [Internet]. 2024 [cited 2025 Jun 12]. Available from: https://www.who.int/news-room/fact-sheets/detail/diabetes
- International Diabetes Federation. Diabetes facts & figures [Internet]. Brussels: IDF; [cited 2025 Jun 12]. Available from: https://idf.org/about-diabetes/diabetes-facts-figures/
- Dang VA, Vu Khanh Q, Nguyen VH, Nguyen T, Nguyen DC. Intelligent healthcare: integration of emerging technologies and internet of things for humanity. Sensors (Basel). 2023 Apr 22;23(9):4200. https://doi.org/10.3390/s23094200. PMID: 37177402; PMCID: PMC10181195.
- Ryalat M, ElMoaqet H, AlFaouri M. Design of a smart factory based on cyber-physical systems and internet of things towards industry 4.0. Appl Sci. 2023;13(4):2156. https://doi.org/10.3390/app13042156
- Mohammed B, Hasan D. Smart healthcare monitoring system using IoT. Int J Interact Mob Technol (iJIM). 2023;17:141–52. https://doi.org/10.3991/ijim.v17i01.34675
- Zhang M, Wei E, Berry R, Huang J. Age-dependent differential privacy. Abstract Proceedings of the 2022 ACM SIGMETRICS/IFIP Performance Joint International Conference on Measurement and Modeling of Computer Systems; 2022. https://doi.org/10.1145/3489048.3526953
- Arefin MS, Rahman MM, Hasan MT, Mahmud M. A topical review on enabling technologies for the Internet of Medical Things: sensors, devices, platforms, and applications. Micromachines. 2024;15(4):479. https://doi.org/10.3390/mi15040479
- Gandrup J, Staniland K, Sharp CA, Dixon WG. Better digital health data should be the foundation to transform outpatient consultations for people living with long-term conditions. J R Soc Med. 2022 Jun;115(6):208–12. https://doi.org/10.1177/01410768221089020. PMID: 35362350; PMCID: PMC9136469.
- Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. 2015;4(1):1. https://doi.org/10.1186/2046-4053-4-1
- Mohammed B, Hasan D. Smart healthcare monitoring system using IoT. Int J Interact Mob Technol (iJIM). 2023;17:141–52. doi:10.3991/ijim.v17i01.34675
- Hussain N, Haddad AHH, Abbas S. Assessing the impact of mobile-based interventions provided by pharmacists on glycemic control and diabetes-related distress in adolescent patients with type 1 diabetes mellitus in Pakistan. Pharm Pract. 2024 [cited 2025 Jun 12]. Available from: https://pharmacypractice.org/index.php/pp/article/view/2946
- Yoo S, Lee DY, Min KP, Park C. Effect of voluntary participation on mobile health care in diabetes management: randomized controlled open-label trial. JMIR Mhealth Uhealth. 2020;8(9):e19153. https://doi.org/10.2196/19153
- Saetang T, Greeviroj P, Thavaraputta S, Santisitthanon P, Houngngam N, Laichuthai N. The effectiveness of telemonitoring and integrated personalized diabetes management in people with insulin-treated type 2 diabetes mellitus. Diabetes Obes Metab. 2024;26(11):5233–8. https://doi.org/10.1111/dom.15870
- Lee EY, Cha SA, Yun JS, Lim SY, Lee JH, Ahn YB, et al. Efficacy of personalized diabetes self-care using an electronic medical record–integrated mobile app in patients with type 2 diabetes: 6-month randomized controlled trial. J Med Internet Res. 2022;24(7):e37430. https://doi.org/10.2196/37430
- Hou M, Qiu W, Qi H, Shao H, Yu J, Bian H. Effects of a diabetes education program integrated with text-message support for lifestyle change among older individuals with type 2 diabetes in communities: a randomised controlled trial. Public Health. 2024;235:152–9. https://doi.org/10.1016/j.puhe.2024.06.032
- Feng Y, Zhao Y, Mao L, Gu M, Yuan H, Lu J, et al. The effectiveness of an eHealth family-based intervention program in patients with uncontrolled type 2 diabetes mellitus (T2DM) in the community via WeChat: randomized controlled trial. JMIR Mhealth Uhealth. 2023;11:e40420. https://doi.org/10.2196/40420
- Dunkel A, Von Storch K, Hochheim M, Zank S, Polidori MC, Woopen C. Long-term effects of a telemedically-assisted lifestyle intervention on glycemic control in patients with type 2 diabetes—a two-armed randomised controlled trial in Germany. J Diabetes Metab Disord. 2023;23(1):519–32. https://doi.org/10.1007/s40200-023-01290-6
- Bouchi R, Izumi K, Ishizuka N, Uemura Y, Ohtsu H, Miyo K, et al. Internet of things-based approach for glycemic control in people with type 2 diabetes: a randomized controlled trial. J Diabetes Investig. 2024;15(9):1287–96. https://doi.org/10.1111/jdi.14227
- Vaughan EM, Hyman DJ, Naik AD, Samson SL, Razjouyan J, Foreyt JP. A telehealth-supported, integrated care with CHWs, and medication-access (TIME) program for diabetes improves HbA1c: a randomized clinical trial. J Gen Intern Med. 2021;36:455–63. https://doi.org/10.1007/s11606-020-06017-4
- Park S, Park JH. Effects of digital self-care intervention for Korean older adults with type 2 diabetes: a randomized controlled trial over 12 weeks. Geriatr Nurs. 2024;58:155–61. https://doi.org/10.1016/j.gerinurse.2024.05.019
- Lee DY, Yoo SH, Min KP, Park CY. Effect of voluntary participation on mobile health care in diabetes management: randomized controlled open-label trial. JMIR Mhealth Uhealth. 2020;8(9):e19153. https://doi.org/10.2196/19153
- Lee Y, Kim G, Jun JE, Park H, Lee WJ, Hwang Y, et al. An integrated digital health care platform for diabetes management with AI-based dietary management: 48-week results from a randomized controlled trial. Diabetes Care. 2023;46(5):959–66. https://doi.org/10.2337/dc22-1929
- Greenwood DA, Blozis SA, Young HM, Nesbitt TS, Quinn CC. Overcoming clinical inertia: a randomized clinical trial of a telehealth remote monitoring intervention using paired glucose testing in adults with type 2 diabetes. J Med Internet Res. 2015;17(7):e178. https://doi.org/10.2196/jmir.4112
- Eghtedari M, Goodarzi-Khoigani M, Shahshahani MS, Javadzade H, Abazari P. Is web-based program effective on self-care behaviors and glycated hemoglobin in patients with type 2 diabetes: a randomized controlled trial. Iran J Nurs Midwifery Res. 2023;28(6):723–9. https://doi.org/10.4103/ijnmr.ijnmr_59_22
- Agastiya IMC, Kurianto E, Akalili H, Wicaksana AL. The impact of telehealth on self-management of patients with type 2 diabetes: a systematic review on interventional studies. Diabetes Metab Syndr. 2022;16(5):102485. https://doi.org/10.1016/j.dsx.2022.102485
- Akingbade O, Nguyen KT, Chow KM. Effect of mHealth interventions on psychological issues experienced by women undergoing chemotherapy for breast cancer: a systematic review and meta-analysis. J Clin Nurs. 2023;32(13–14):3058–73. https://doi.org/10.1111/jocn.16533
- Akingbade O, Hong KY, Ayo OS, Alade AS, Lawal MA, Somoye IE, et al. Development and feasibility of an mHealth intervention for psychoeducational support of Nigerian women diagnosed with breast cancer undergoing chemotherapy: a pilot randomized controlled trial. PLoS One. 2024;19(11):e0314365. https://doi.org/10.1371/journal.pone.0314365
- Akingbade O, Ayo O, Alade A, Lawal M, Eze C, Chow KM, et al. Effect of a nurse-led mHealth psychoeducational intervention on self-efficacy, coping and psychological distress of Nigerian women with breast cancer receiving chemotherapy: a randomized controlled trial. Oncol Nurs Forum. 2024;51(2):E2319–20.
- Fortmann AL, Gallo LC, Garcia MI, Taleb M, Euyoque JA, Clark T, et al. Dulce Digital: an mHealth SMS-based intervention improves glycemic control in hispanics with type 2 diabetes. Diabetes Care. 2017;40(10):1349–55. https://doi.org/10.2337/dc17-0230
- Akingbade O, Adediran V, Somoye IE, Alade AS, Chow KM. Perceived feasibility and usefulness of mHealth interventions for psychoeducational support among Nigerian women receiving chemotherapy for breast cancer: a focus group study. Support Care Cancer. 2022;30(12):9723–34. https://doi.org/10.1007/s00520-022-07403-w
- Guo M, Meng F, Guo Q, Bai T, Hong Y, Song F, Ma Y. Effectiveness of mHealth management with an implantable glucose sensor and a mobile application among Chinese adults with type 2 diabetes. J Telemed Telecare. 2021;29(8):632–40. https://doi.org/10.1177/1357633x211020261
Copyright Ownership: This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, and the use is non-commercial. See http://creativecommons.org/licenses/by-nc/4.0.
Appendix
| Study number, citation, country, and study duration | Number demographics, mean age, and BMI | Intervention (components, format, intervener, platform, duration, frequency, and dosage). | Control group | Outcome measures and instruments for measurement | Effect of intervention. Theoretical basis |
| 1 Addie et al., 2017, San Diego, California 6 months |
N = 126 Male and female Mean age: 48.43 years |
Dulce Digital (participants received up to three motivational, educational, and/or call-to-action text messages per day over 6 months on cell phones). | Received UC | HbA1c, TC, LDL TC, HDL-C, and triglycerides) measured by the laboratories of Quest Diagnostics (West Hills, CA). Systolic/diastolic BP measured with a standardized protocol according to guidelines using a standard digital sphygmomanometer (HEM-907XL; Omron). Body weight/height measured using a traditional balance scale and stadiometer to the nearest 0.1 lb and 0.2 inch. |
Dulce Digital group exhibited a significantly lower mean HbA1c compared with UC at month 3 (8.5 ± 1.2% [69.0 ± 13.1 mmol/mol] vs. 9.3 ± 1.9% [78.0 ± 20.8 mmol/mol], P = 0.03) and month 6 (8.5 ± 1.2% [69.0 ± 13.1 mmol/mol] vs. 9.4 ± 2.0% [78.0 ± 20.8 mmol/mol], P = 0.03), after controlling for baseline HbA1c. Theoretical basis: None. |
| 2 Shahid et al., 2015, Pakistan 4 months |
N = 440 Male and female, Mean age: Intervention: 48.95 years. Control: 49.21 years. Mean BMI: Intervention: 27.08 kg/m² Control: 27.57 kg/m² |
SMBG form was given to monitor. BG levels and regular (15 days) feedback based on their BG over the past readings of 15 days were received on their phone. |
SMBG form was given to monitor. BG levels with no feedback based on their BG. |
Each patient received 8 calls during the entire study. Patients were asked about the SMBG, intake of medications, physical activity, and healthy eating. |
Theoretical basis: None. |
| 3 Feng et al., 2023 China 12 months |
N = 225 Male and female Mean age: 65.6 years |
Online intervention: Implementation included three phases. Phase 1: family members were asked to follow the WeChat account and add it to their personal WeChat. Phase 2: study personnel regularly delivered 38 articles (~3 articles were delivered per week (Monday, Wednesday, and Friday). All online intervention articles were delivered at 6 AM or 5 PM on ‘push day’ (assigned day) so that participants could receive the intervention outside working hours. Phase 3, effectiveness of online intervention was measured. |
Control group received on-site health education once every 3 months. | HbA1c was the primary outcome. | Lower HbA1c values (β = –0.69, 95% CI: −0.99 to −0.39; P < 0.001) and improved scores for general diet (β = 0.60, 95% CI: 0.20–1.00; P = 0.003), special diet (β = 0.71, 95% CI: 0.34–1.09; P < 0.001). BG testing (β = 0.50, 95% CI: 0.02–0.98; P = 0.04) KAP model and HBM. |
| 4 Hussain et al., 2024, Pakistan 6 months |
N = 112 Male Mean age: 15.2 years |
5 pre-selected glucose tracking apps available in the Urdu language were downloaded for the MTG. Information on SH and DK were collected directly from the study participant and from data extraction from the medical record. Discrepancies between the 2 sources could be connected to underreporting in the medical records and overreporting by the study participants and their families, particularly for SH. |
RTG was treated with the standard hospital and treating physician protocol without involvement of mobile app use. RTG participants met with the pharmacist to have demographic data collected only. |
HbA1c levels. Episodes of severe DKA. Diabetes distress levels. |
Group RTG versus Group MTG exhibited higher HbA1c (9.2 vs. 7.5). The difference in HbA1c between the 2 groups was 1.8%, and statistically significant. RTG had a higher number of severe hypoglycemic (9±2.0 vs. 5±1.4, p 0.05), and DKA episodes (6±1.8 vs. 3±2.1, p = 0.02) in 12 months compared to MTG in 12 months at the end of the study. Theoretical basis: None. |
| 5 Lee et al., 2023, Korea 48-week |
N = 294 Male and female Mean age: 56.11 years Mean baseline HbA1c in the FAS: 7.45% |
Experimental Group B used the digital integrated health care platform without medical supervision. Medical staff helped experimental Group C install the platform on their mobile devices and instructed them on its use and use at the hospital. Groups B and C got Bluetooth-connected glucometers, sphygmomanometers, scales with bioelectrical impedance analysis, and watch-type pedometers. |
Routine diabetes care with hospital visits every 3 months was applied for control Group A. | HbA1c levels using a digital integrated health are platform and mobile devices. | Groups B and C showed significantly larger decreases in HbA1c from baseline to 24 and 48 weeks (-0.32 ± 0.58% and -0.28 ± 0.56%, respectively) vs. Group A (-0.06 ± 0.61% and 0.07 ± 0.78%). Groups B and C lost more weight than group A from baseline to 24 weeks and 48 weeks, respectively. |
| 6 Dunkel et al., 2024 Germany 12 Months |
N = 151 Male and female Mean age: 59.7 years. |
Each participant received a tablet PC, pedometer, and blood glucose meter for telemonitoring. All devices were connected via Bluetooth, which automatically transfers data from the glucometer and pedometer to the tablet computer. The diabetes coach supervised the patient over a year. The devices motivate and provide continuous feedback. |
Control group received UC. | HbA1c levels were recorded by the patients’ physicians and served as primary outcomes in this study. Secondary outcomes were BMI, physician contacts, costs for antidiabetics, physical activity and use of technology. |
At the end of the intervention (-0.52 (-0.73; − 0.32), p < 0.000) and the 24-month follow-up (-0.38 (-0.61; − 0.15), p < 0.001), the intervention group showed significantly better HbA1c values compared to the control group. |
| 7 Park & Park, 2024, Korea 12 weeks |
N = 120 Male and female Mean age: 72.70 years; Mean BMI: 24.13 ± 3.09. |
Standardized messages from DiaNote app promoted self-care and risk awareness. If data were not entered for 3 days or 1 day, these reminders were sent daily at 8:00 pm. When BG exceeded 250 mg/dL for 3 days, real-time reminders were sent. This proactive strategy relied on notification services. |
Traditional logbook containing details on diet, exercise, medication, and BG levels. | Diet, exercise, medication adherence, and BG levels were recorded using the DiaNote app. | The DiaNote group’s HbA1c reduction was the DSI program’s main result. This showed that a 12-week app-based DSI intervention improved HbA1c levels more in the DiaNote group than in the logbook group. Theoretical basis: None. |
| 8 Lee et al., 2022, Korea 6 months |
N = 234 Male and female, mean age: 52.5 years Mean BMI: 27.2 kg/m2 Mean baseline HbA1c: 8.7% Mean duration of diabetes: 11.4 years |
MC and MC with MPC intervention groups used a diabetes self-care mobile app and UC to treat diabetes. The app recorded patient SMBG, diet, and step count) and sent automated text messages (educational, behavioral, and motivational) from the iCareD system to their phones. Two standardized diabetes self-care and lifestyle modification messages and a lifestyle questionnaire-specific message were sent 3 times a week. |
Control received UC per the Korean Diabetes Association standard care for T2DM patients. | The primary outcome compared the change in HbA1c levels (%) between baseline and 26 weeks for the 3 groups. The secondary result compared HbA1c and FBG levels (mg/dL) between the UC and two mobile-based intervention groups from baseline to 26 weeks. |
No significant difference in HbA1c values across the three groups at 26 weeks. At 12 weeks, the 3 groups had significantly differing HbA1c reductions. The post hoc analysis showed that only the MPC group had a significant HbA1c decline compared to the UC group. Theoretical basis: None. |
| 9 Eghtedari et al., 2023, Iran 7 months |
N = 70 Male and female |
The study involved a web-based program that included multimedia educational content (e.g. videos, lectures, educational motion graphics, text files, posters, and podcasts) presented in 7 sections based on DSMES over a 21-day period with monitoring by an instructor. | Control group was provided with educational pamphlets. | Fasting blood samples (2.50 ml) were collected before and after the interventions to measure HbA1c levels. | After intervention, mean scores for healthy eating and medication adherence significantly increased in the intervention group, and the mean differences in HbA1c values significantly decreased in the intervention group compared to the control group. Theoretical basis: None. |
| 10 Lee, et al; 2022 Korea 26 weeks |
N = 269 Male and female Mean age 52.5 years Mean BMI: 27.2 kg/m2 |
MC and MPC groups.DMP (iCareD application, Medical Excellence Inc., Seoul, Korea) is used in addition to regular care. The app allows patients to enter self-care data, SMBG, dietary habits, and step count), and automated text messages (educational, behavioral, and motivational messages) from the iCareD system were sent to their mobile phones. Automated messages were sent 3 times per week and consisted of 2 standardized messages for diabetes self-care and lifestyle modification and a customized message according to the lifestyle questionnaire. In the iCareD system, specific content for each message was developed based on the clinical practice guidelines of the Korean Diabetes Association and multidisciplinary expert opinions from the diabetes care team (endocrinologist, certified dietician, and diabetes educator). Mobile app was integrated to hospital EMR. A health care provider sent additional recommendations and bidirectional feedback to each participant every 2 weeks through the iCareD system during the intervention period. Feedback was mainly related to diabetes self-care, the SMBG, or lifestyle modification. Patients physically visited the outpatient clinic every 3 months. |
Provision of glucometer, basic diabetes education, including information on SMBG, diet, and PA. The usual standard care for patients with T2DM by the Korean Diabetes Association. |
The primary outcome was the difference in the change in HbA1c (%) between baseline and 26 weeks among groups. The secondary outcome was the changes in HbA1c and FBG (mg/dL) between the UC and 2 mobile-based intervention groups between baseline and 26 weeks every 26 weeks. |
At 12 weeks after intervention, the mean decline in HbA1c was significantly different among the 3 groups. HbA1c showed a statistically significant decrease after the intervention in the MPC group vs. the change in the UC or MC group, especially in patients aged <65 years (P = 0.02), patients with a diabetes duration of ≥10 years (P = 0.02), patients with a BMI of ≥25.0 kg/m2 (P = 0.004), patients with a C-peptide level of ≥0.6 ng/mL (P = 0.008), and patients who did not undergo treatment with insulin (P = 0.004) at 12 weeks. Theoretical basis: None. |
| 11 Saetang, et al; 2024 Thailand 6 months |
N = 151 Male and female Mean age 53.36 years |
Intervention group (tele-iPDM) received insulin adjustments by investigators through a cloud-based telemonitoring platform for 6 months (blood glucose monitoring reviewed weekly from Weeks 0 to 12 and then monthly from Weeks 13 to 24). | Control performed self-monitoring and insulin adjustment. | The primary outcome was the difference in HbA1c change from baseline between the 2 groups at 24 weeks. Secondary outcomes included changes in HbA1c at 12 weeks, FBG, body weight, BMI, the percentage of individuals achieving HbA1c <7% at 24 weeks, the percentage of individuals with an HbA1c reduction of >0.5% at 24 weeks, and incidences of hypoglycemic events. |
The mean difference in HbA1c between the tele-iPDM and usual care groups was −0.65% (95%CI: −0.99 to −0.30; p < 0.001). No significant differences in body weight, BMI, or hypoglycemic events between the two groups. |
| 12 Yoo, et al; 2020 Korea 6 months |
N = 66 Male and female Control: Mean age 53.2 years, 25.5 kg/m2 Intervention: Mean BMI 50.0 and 26.8 kg/m2 |
Participants in the intervention group received mHealth-based diabetes self-management education consisting of the mobile app Healthynote (for Android; CVnet Co) and regular individualized feedback messages from healthcare professionals regarding their diabetes management. Intervention group uploaded the Healthynote app to their own smartphone, were educated on how to use this service, and were provided a near field communication–enabled glucometer (CareSens N NFC; i-SENS Inc). During the 6-month study, participants in the intervention group received regular mobile messages from healthcare professionals, consisting of 2 endocrinologists and a nurse, via the mobile app once or twice a week. |
Control maintained previous diabetes management at Kangbuk Samsung Hospital. | The primary outcome was change in glycated hemoglobin (HbA1c) level over 6 months between the 2 groups (intervention vs. control) and within each group (at 6 months vs. baseline). The secondary outcomes were changes in body mass index, blood pressure, lipid profile, and questionnaire scores (the Korean version of the Summary of Diabetes Self-Care Activities Questionnaire, an Audit of Diabetes Dependent Quality of Life, the Appraisal of Diabetes Scale, and Problem Areas in Diabetes) over 6 months between groups and within each group. | mHealth-based diabetes self-management education was effective at improving glycemic control and diabetes self-management skills and lowering diabetes-related distress in voluntary participants. Theoretical basis: None. |
| 13 Greenwood, et al; 2015 California 10 months |
N = 90 Male and female Mean age: 58 years Mean BMI: 34.1 kg/m2 |
Participants in this group attended a 1-hour, small group, in-person training session led by the CDE that included 1) use of the glucose meter, 2) implementation of the complete feedback loop, 3) use of paired glucose testing (frequency and intensity of monitoring), 4) ADA goals for pre- and postmeal, 5) how to use SMBG data to modify behavior or treatment, 6) expected feedback from CDEs with communication by secure message or phone, 7) the use of shared decision making to implement the treatment plan. During training, participants learned how to use the Care Innovations Guide, a telehealth remote monitoring system approved by U.S. FDA, which includes an in-home tablet computer connected by Internet or 3G network to the Care Innovations Health Suite online portal. |
Usual care received diabetes education booklets and referral for formal diabetes education as needed. This group continued receiving nurse care coordination, including reminders for HbA1c and health maintenance exams sent by mail. | The primary outcome was the difference in mean change in the HBA1c from baseline to 6 months between groups. Secondary outcome included diabetes knowledge, self-management, and self-efficacy. |
An eHealth model incorporating a complete feedback loop with telehealth remote monitoring and paired glucose testing with asynchronous data analysis significantly improved HbA1c levels compared to usual care. Theoretical basis: None. |
| 14 Guo, et al; 2021 China 4 weeks |
N = 68 Male and female Control: Mean age 59.46 years. ,BMI 24.32 kg/m2 Intervention: 55.25 years, BMI: 24.00 kg/m2 |
Patients in the intervention group received the mHealth management model consisting of the network platform, an implantable glucose sensor, a mobile app, and GP support. | The control group received usual health management. | The indicators used to evaluate the effectiveness of intervention included BMI, FBG, postprandial 2-hour BG (2hPG), HbA1c, quality of life, and self-management ability. | mHealth management showed significant reductions in BMI, FBG, 2h PG, HbA1c, and improved quality of life and self-management among patients, which may be related to real-time feedback from an implantable glucose sensor and guidance from GPs through a mobile app. mHealth management is a very promising way to promote the health management of T2DM in China, and this study provides a point of reference for mHealth management abroad. Theoretical basis: None. |
| 15 Bouchi, et al; 2024 Japan 52 weeks |
N = 1,159 Male and female Mean age 56.2 Mean BMI 26.1 kg/m2 |
This group was supervised to use an IoT automated system that presents a summary of lifelogging data (weight, BP, and physical activities), and provides feedback messages twice weekly that promote behavioral changes in diet and exercise. | Usual care provided with devices with IoT functions. | The primary end point was a HbA1c change over 52 weeks. Key secondary endpoints were intergroup differences in weight, BMI, SBP, and DBP, plasma glucose, TC, HDL-C, ALT, creatinine, intragroup changes in scores obtained from the BDHQ from baseline to 52 weeks, and the DTR-QOL from baseline to 24 and 52 weeks and the end of the study. |
IoT-based approach did not reduce HbA1c in patients with T2DM. Theoretical basis: None. |
| 16 Vaughan, et al; 2021 Texas 12 months |
N = 89 Male and female Control Mean age: 53.86 years Mean BMI: 34.56 kg/m2 Intervention Mean age: 55.99 years Mean BMI: 32.59 kg/m2 TIR group |
Participants in this group received the TIME intervention consisting of 1) CHW-participant telehealth communication via mobile health (mHealth) for 12 months, 2) CHW-led monthly group visits for 6 months, and 3) weekly CHW-physician diabetes training and support via telehealth (video conferencing). | Participants received usual care. | The primary study outcome was glycemic control as measured by baseline to month 6 change of HbA1c. Therapeutic clinical outcomes were BP, weight, and BMI. | TIME participants resulted in improved outcomes, including HbA1c. Theoretical basis: None. |
| 17 Hou, et al; 2024 China 6 months |
N = 105 Male and female Control Mean age 64.5 years Mean BMI 24.8 kg/m2 Intervention 69.2 years Mean BMI 24.6 kg/m2 |
Intervention group received 6-month DSME and text-message support for behavior change (self-management education integrated with text message support; SME-TMS) in addition to usual care. | The control group performed self-monitoring and insulin adjustment. | The primary outcome was reductions in HbA1c and FBG at 6-month nonintervention follow-up. Secondary outcomes were reductions in body weight, BMI, BP, TC, LDL-C, HDL-C, physical activity, and health beliefs. |
Intervention led to substantially increase in days of weekly physical activity (42% vs. 0%, P < 0.001) and health beliefs (coefficient = 7.0, 95% CI: 4.4 to 9.6, P < 0.001). But no greater reduction was found in HbA1c at 6 months after the intervention, compared with the control group (0.13%, 95% CI: −0.20 to 0.46, P = 0.443). Reductions of blood pressure, TC, and LDL-C were greater in the control group than in the intervention group (all P < 0.050). In the intervention group, participants had a significant reduction in BMI. The control group had greater reductions in TC and LDL-C (all P < 0.050). Theoretical basis: None. |
| ADA, American Diabetes Association; ALT, alanine transaminase; BMI, body mass index; BG, blood glucose; BP, blood pressure; BDHQ, brief-type self-administered diet history questionnaire; CDE, certified diabetes instructor; CHW, community health worker; CI, confidence interval; DBP, diastolic blood pressure; DMP, Diabetes Management Program; DSME, diabetes self-management education and support; DTR-QOL, Diabetes Therapy-Related Quality of Life; DK, diabetic ketoacidosis; DKA, diabetes ketoacidosis; FBGL, fasting blood glucose; FDA, Food and Drug Administration; GP, general practitioner; PDM, integrated personalized diabetes management; FBG, fasting blood glucose; HBM, Health Belief Model; HDL-C, high-density lipoprotein cholesterol; IoT, Internet of Things; KAP, knowledge-attitude-practice; lb, pound; LDL-C, low-density lipoprotein cholesterol; MC, mobile diabetes self-care; MPC, personalized; bidirectional feedback from physicians; MTG, Medical Treatment Guidelines; PA, physician assistant; PC, personal computer; RTG, routine treatment group; SBP, systolic blood pressure; SH, severe hypoglycemia; SMBG, self-monitoring blood glucose; SME-TMSL, self-management education and text-message support; T2DM, type 2 diabetes mellitus; TC, total cholesterol; TIME, time in range; UC, usual care. | |||||