NARRATIVE/SYSTEMATIC REVIEWS/META-ANALYSIS
The Impact of Telemedicine on the Pharmaceutical Industry in Brazil
Shirley Meschke Mendes Franklin de Oliveira, JD and LLM, Law, PhD, Business Administration
Pfizer Legal Division. This work was conducted by the author at Sindusfarma, while studying at FGV-EAESP (Fundação Getúlio Vargas – Escola de Administração de Empresas).
Keywords: Business model digital health, pharmaceutical industry, telehealth, telemedicine
Abstract
This article analyzes the impact of telemedicine on the pharmaceutical industry of the Pharmaceutical Products Industry Union of the State of São Paulo, Brazil (Sindusfarma). The source material used is the result of monitoring the work by the Digital Health Committee, including analysis of the work and discussions related to marketing and business support, regulatory and legal strategy, and access committees, as well as interviews with industry representatives. The global and Brazilian digital health strategies and the pharmaceutical industry’s business model are evaluated to identify the impact of telemedicine on the business model. An opportunity map is presented to expand the practice of telemedicine with suggestions for actions for pharmaceutical companies and project considerations for Sindusfarma. The goal is to contribute to the Digital Health Strategic Plan, innovate and modernize the sector, and increase patient access to information, prevention, diagnosis, and treatment.
Citation: Telehealth and Medicine Today © 2025, 10: 589
DOI: https://doi.org/10.30953/thmt.v10.589
Copyright: © 2025 The Authors. This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0. The authors of this article own the copyright.
Submitted: May 13, 2025; Accepted: September 3, 2025; Published: September 30, 2025
Corresponding Author: Oliveira, Shirley Meschke M Franklin de, Email: Shirley.Meschke@pfizer.com
Competing interests and funding: The author reports no conflicts of interest. The author discloses having an employment relationship with the Legal Division of Pfizer Pharmaceuticals. However, this article is an independent without any interference or influence from Pfizer, and it relies solely on public records, interviews, and documents shared by Sindusfarma, where the participant’s research was conducted.
None
This article aims, from the perspective of Sindusfarma, to present a brief analysis of the changes brought about by telemedicine and its impacts on the pharmaceutical industry and contribute to the expansion of patient access strategies to treatments, increased awareness of disease prevention through the collaboration of all members of the organizational field, and maximization of technology availability focused on the sustainability of the health system.
In 2020, the World Health Organization (WHO) highlighted the 13 greatest health challenges for the following decade: 1) keeping health care clean; 2) elevating health in the climate; 3) delivering health in conflict and crisis; 4) making health care fairer; 5) expanding access to medicines; 6) stopping infectious diseases; 7) preparing for epidemics; 8) protecting people from dangerous products; 9) investing in health workers; 10) keeping adolescents safe; 11) earning public trust; 12) harnessing new technologies; and 13) facilitating the appropriate use of medications.1
Healthcare in Brazil faces the same challenges, especially the need to guarantee citizens’ access to information about disease prevention and treatment, as well as effective treatments. Law 8.080/1990, in its second article, states that health is a fundamental right, and the state must provide the conditions for its application. The state has a duty to formulate and implement policies aimed at reducing the risk of diseases and other health issues and establish conditions that ensure universal and equal access to actions and services for the promotion, protection, and recovery of health.
The sustainability of health systems, in general, depends on the articulation and collaboration of all participants in the health organizational field. The health sector is understood to include pharmaceutical industries, distributors, service providers, health insurance companies and plan operators, patients, physicians, suppliers of inputs and services, the government, and the population as a whole.
Given the relevance of the topic, the Pharmaceutical Products Industry Union in the State of São Paulo (Sindusfarma) was selected as the workplace and case study site. Sindusfarma includes national and international pharmaceutical companies of all sizes and specialties, as well as suppliers and service providers operating in the sector, and accounting for over 95% of the pharmaceutical market in the country, allowing for a broad understanding of the dynamics involved in the business model transformation of associated pharmaceutical industries in light of digital transformations.
Digital transformation is understood here as the digital acceleration of healthcare, specifically the expansion and sophistication of telemedicine as a practice to help meet the diverse needs of patients. The digital transformation has led to the emergence of new organizations and business models within the healthcare sector. Changes and adaptations to the digital environment must be swift and strategic to provide patients with greater access to information, increased awareness about diseases, prevention, and access to treatments, as well as to optimize resources dedicated to healthcare.
This transformation has also been driven by the needs, experiences, and opportunities arising from the COVID-19 pandemic, for example, the authorization of telemedicine use for consultations between doctors and patients, pharmaceutical product sample deliveries by delivery companies, electronic prescriptions, and the increase in online courses and events, among others. This has led to a new perspective on the external context and the strategies of organizations.
The business model of the industry must serve the entire healthcare ecosystem, defined by Arthur Boni as the 5Ps (Physicians; Providers; Payers; Patients; Partners).2 According to the author, digital transformation requires greater collaboration among industries to achieve the convergent goal of offering accessible therapies that create value for all parties, including the government and the general public.
The definition of a business model is not consolidated in the literature. Joan Magretta3 understands it as the “story that explains how the business operates” and says that a good business model answers questions such as “Who is the customer and what does the customer value?” and “What economic logic explains how to deliver value to the customer at an appropriate cost?”. Osterwalder, Pigneur, and Tucci4 describe it as a conceptual tool that contains a set of elements and their relationships, allowing the expression of a company’s business logic. Casadesus-Masanell and Ricart5 define it as the way organizations achieve profit by addressing two fundamental aspects: how they identify and create value for customers and how they capture part of this value as profit in the process. Zott, Amit and Massa6 understand it as the content, structure, and governance of transactions intended to create value through the exploitation of business opportunities.
The definition of created value requires an external analysis of what guides the consumers to whom the product or service is intended and the interests of the suppliers.7 The processes of value creation and capture are distinct, as the one who creates the value may or may not be able to capture or retain it in the long term.8
The research of new therapies aims to meet the current, imminent, or future treatment needs of patients, to seek prevention, and to enable early diagnosis of diseases. From the perspective of the pharmaceutical industry, this research purpose may already represent the value offered to patients. In addition to research, the manufacturing industry aims to produce medications and therapies with quality, in order to deliver to potential patients, the benefits identified through research. In this sense, the rigor of the manufacturing process could also be understood as value for the patient. This would be understood as value creation.
The way medications are marketed is also critical to maintaining product quality and delivering value, as it requires commitment and responsibility from various partners in the distribution chain, as well as compliance with regulatory legislation. The dissemination of information from clinical studies, medications, the need for early diagnosis, and awareness of certain diseases to the medical category and healthcare professionals, who mediate relationships between research industries, manufacturers, and patients, is equally relevant. Healthcare professionals need continuous education due to the rapid advancement of research and discoveries, and this is provided through industry representatives, researchers, and doctors at educational scientific events.
Industries also have patient programs through which relevant information about diseases, and their prevention is disseminated, and support is provided for the proper administration of prescribed medications. This is the most direct channel of interaction with the patient, where the industry can identify elements of the value delivery, such as satisfaction with the product and service, any adverse events, and adherence to treatment, for example. Thus, the forms of marketing and dissemination can be understood as Value Delivery. The capture of the value created and delivered by the pharmaceutical industry occurs with the patient’s adherence to treatment, the choice of therapy or medication by healthcare professionals, and the increased awareness of patients about prevention, diseases, and available treatments.
Digital Health in Brazil
The healthcare in Brazil sector has never been simple and is becoming increasingly challenging. New technologies have accelerated innovation, new competitors are emerging from different parts of the world, and customers are becoming more sophisticated and more demanding.9
With the COVID-19 pandemic, barriers to physical visits led to the creation of alternatives in the healthcare field. The pharmaceutical industry, which previously delivered free medication samples through its sales representatives, began to do so via specialized companies.10,11 Many continuing education courses began to be offered virtually, expanding access for healthcare professionals to medical and scientific events.
Regarding customers, the use of data analysis technology allows for more efficient business management, in addition to enabling the implementation of risk-sharing contracts (based on performance/efficacy, fixed-price product portfolio purchases per patient, for example), which rely on data analysis. New technologies are entering the healthcare services ecosystem, driven by companies that transcend geographic, cultural, and regulatory boundaries.12 These newcomers to the healthcare sector bring opportunities for new businesses and might change the dynamics of relationships. This transformation in the sector must be accompanied by collaboration among all members of the organizational field of healthcare, aiming at the sustainability of the health system.
Table 1 listed the projects of Sindusfarma and its connection with the proposal made to the government. Sindusfarma goals broad: “1) to foster relationships between entities in the pharmaceutical industry, healthcare professionals, and those in medical and pharmaceutical sciences; 2) to defend the interests of the economic category of the industry, importers, and exporters of pharmaceutical products, related goods, functional foods, or any other product with prophylactic, curative, palliative, or diagnostic purposes also registered with the Brazilian Health Regulatory Agency (ANVISA); 3) to collaborate with public authorities and other associations in the spirit of social solidarity and subordination to national interests; 4) to define regulatory frameworks; and 5) to introduce and strengthen the concepts of Good Manufacturing Practices”.13
In Brazil, in 2020, the Strategic Vision for Digital Health 2020–2028 was defined, outlining seven priorities, relevant stakeholders, and the type of expected participation. The strategic vision seeks to consolidate the work carried out over the past decade, which has been formalized in documents such as the National Policy on Health Information and Informatics,13 the e-Health Strategy for Brazil,14 and the Action, Monitoring, and Evaluation Plan for Digital Health in Brazil (2019–2023).15
According to the Strategic Vision of the Ministry of Health in Brazil, the essential characteristics of digital health can be summarized in the following statements: 1) the best strategies are led by the public sector and are inclusive, open, and participatory; 2) it is urgent today that innovation, knowledge, and best practices developed in any of the sectors related to health be rapidly tested, validated, and implemented; 3) it is evident that the public sector alone cannot meet all the needs for scientific discovery, industrial production, innovation, and human resource training for health, and even less so for digital health; 4) there is significant lack of understanding and a high level of distrust between public and private sector actors, both in healthcare and in other industries related to supplies, equipment, services, and health products; and 5) digital health is an extremely complex area in terms of knowledge and practice, due to the diversity of actors and interests, the immaturity of healthcare organizations, the shortage of human resources and qualified leadership, and, above all, the inherent complexity of healthcare processes.16
On January 1, 2023, through Decree No. 11.358, the Secretariat of Information and Digital Health (SEIDIGI) was created within the Ministry of Health.17 The first step established is the formation of an integrated and collaborative effort among all stakeholders involved in the process. The second is the digitization of the three levels of care, with the goal of promoting the implementation of digital health system policies and accelerating the adoption of electronic medical records and hospital management systems as integral parts of healthcare processes. The third is support for the improvement of healthcare, ensuring that the National Health Data Network (RNDS) supports best clinical practices through services such as telehealth and apps developed by the Ministry of Health or its partners. By 2028, the aim is to engage patients and citizens to encourage the adoption of healthy habits and the management of their own and their families’ health. It is expected that citizens will take a more active role and help build the digital health systems they will use. The training and education of healthcare professionals in informatics is also a priority, which will ensure recognition of health informatics as a field of research and health informaticians as a profession.18
Creating an interconnected environment is also a priority. The RNDS will enhance collaborative work across all sectors of healthcare so that technologies, concepts, standards, service models, policies, and regulations can be put into practice. Finally, it aims to establish an innovation ecosystem. An innovation lab has been created to develop guidelines and foster new ideas based on the needs of the Unified Health System (SUS).18
Methodology
The research methodology used was participant research, with a qualitative, exploratory-descriptive approach, conducted within Sindusfarma.
The research was divided into three phases: investigation, thematization, and programming. Before beginning the investigation phase, a literature review was conducted, gathering academic papers and other texts related to telemedicine, digital marketing, innovative partnerships, innovation, strategy, business models, and competitiveness in management. This review contributed to the conclusions drawn during the investigation phase and later supported the thematization process.
During the investigation phase, basic information about the field of work was collected. Meetings of the Digital Health Committee were observed, and the minutes from meetings of the Marketing and Business Support, Regulatory, Legal, and Access Committees, when they included items on the agenda related to digital transformation, were requested for analysis.
To better diagnose the challenges and opportunities of telehealth in the country, the Digital Health Committee organized a public meeting with the main objective of discussing how telehealth can contribute to improving healthcare, one of the priorities of the Digital Health Strategic Plan.
The group consisted of pharmaceutical industries and service providers, and the event was conducted online on March 14, 2023. The event’s design, facilitation, and analysis were carried out by the Sindusfarma Committee. The key discussion points were: 1) the history, complexity, and regulation of telehealth; 2) the challenges and opportunities of telehealth, and how it can contribute to improving healthcare; 3) the digital acceleration of modern society and the digital modernization of healthcare; 4) the distinction between ‘telehealth’ and ‘telemedicine’, and the importance of this differentiation to avoid ethical and legal issues; 5) how telehealth can contribute to the sustainability of the healthcare system; 6) important strategies that can be adopted by the pharmaceutical industry in engaging partners resulting from the possibilities arising from digital advances.
Still in the diagnostic phase, interviews were conducted with: 1) the President of a patients’ association for rare diseases, to understand the difficulties related to access to information, prevention, and innovative therapies from the patient’s perspective, as well as the opportunities and challenges of telemedicine; 2) a former Secretary of the Ministry of Health, to understand the priorities, challenges, and expectations regarding the sustainability of the national healthcare system; 3) the President of Sindusfarma, to understand the impact of digital transformation in the health sector, particularly in the pharmaceutical industry; and 4) the President of a digital solutions company that developed a digital health ecosystem connecting doctors, patients, and pharmacies, in partnership with hospitals, health insurers, pharmacies, software companies, and the pharmaceutical industry. The interviewees were selected because each represents a different organization within the sector that interacts with the pharmaceutical industry and offers complementary perspectives.
The most relevant themes identified through participation in the meetings of the Sindusfarma Digital Health Committee, the analysis of minutes from other committees, the 2022–2026 business project, and the interviews were classified and prioritized. Subsequently, a map was created with suggestions for potential action projects that Sindusfarma may later implement with its member companies.
Investigation
Proposal to Improve Access to Medicines Presented to the Government by Sindusfarma
Table 1 presents a summary of the main proposals submitted by Sindusfarma to Luiz Inácio Lula da Silva’s government and its business project as an entity for the 2023 to 2026, demonstrating the organization’s strategic and active role in addressing relevant issues in the sector.
Participation in the Sindusfarma Digital Health Committee and the Strategic Digital Health Plan of Brazil’s Ministry of Health
The official launch meeting of the Sindusfarma Digital Health Committee, in partnership with the Brazilian Network for Pharmaceutical Innovation, took place on November 23, 2022. The main goal of the committee was to discuss and increase the level of knowledge among Sindusfarma’s members and society in general about the importance of the RNDS, bringing analysis, discussion, and action proposals related to the following pillars of the Digital Health Strategy: support for improving healthcare, the user as the protagonist, and the innovation ecosystem. The target audience included professionals working in the field of digital health, both within and outside the pharmaceutical industry.19
The topic of Interoperability was discussed, as established by Ordinance No. 2.073/2021 of the Ministry of Health, which regulates the use of health information interoperability standards within the Unified Health System (SUS), across municipal, district, state, and federal levels, as well as in private and supplementary health systems.
According to Brazil’s Digital Health Strategy, by 2028, the RNDS will be established and recognized as the digital platform for innovation, information, and health services across Brazil, for the benefit of users, citizens, managers, professionals, communities, and health organizations. The goal of the RNDS is to create a repository for the exchange of information across the various levels of healthcare, enabling continuity and transition of care in both public and private sectors.
The Strategic Digital Health Plan for Brazil and its connection with the priorities of the National Policy on Health Information and Informatics were discussed. The plan established seven priorities (Figure 1). The committee chose to focus on three of them in which Sindusfarma and its members can make the most significant contribution: 1) support for improving healthcare; 2) the user as the protagonist; and 3) the ecosystem and innovation. The strategic plan of the Ministry of Health is aligned with the guidelines set forth in the World Health Organization’s Global Digital Health Strategy.
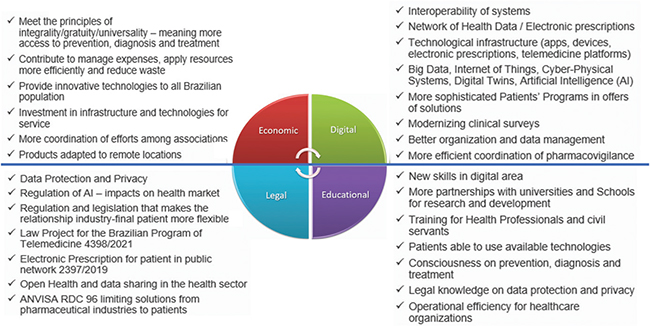
Fig. 1. Opportunities to maximize the use of telemedicine to boost the revision of the business model for more competitiveness and achieve the goals of the pharmaceutical industry. AI: artificial intelligence; ANVISTA: The Brazilian Health Regulatory Agency; RDC 96: Collegiate Board Resolution (Resolução da Diretoria Colegiada).
Source: Elaborated by the author.
One of the key elements supporting improvements in healthcare is system interoperability, which is the ability of two or more systems or components to exchange and use information. This is directly linked to the innovation ecosystem, which still needs to be developed.
Another relevant topic is security (Table 2), which should be addressed under the priority of ‘digitization of the three levels of care’, comprising 1) expansion of internet connectivity, 2) expansion of digitization in primary care, and 3) expansion of digitization in other levels of care. Given the importance of this topic, one possibility could be to establish a separate priority or pillar to address security. Understanding the strategic actions of the Department of Information and Informatics of Brazil’s Unified Health System (DataSUS) is important to identify opportunities for partnership and collaboration. The Strategic Plan’s design did not involve all potential collaborators, making this an opportune moment for those with something to contribute to engage. The pharmaceutical industry was not involved in this process, which led to the creation of the Digital Health Committee within Sindusfarma.
The ‘Innovation Ecosystem’ priority may need to be expanded to include the expectations and contributions of other collaborators and partners. The National Health Data Network will be a major platform that will serve as the national interoperability channel for exchanging health data. The several DataSUS systems need to be interconnected. The electronic patient record is also a relevant priority.
The Ministry of Health already has ConecteSUS, an application that records the entire trajectory of individuals seeking care through SUS.14 It displays, for example, data on patient visits and hospitalizations. It allows users to view prescribed medications, complete exams, and schedule appointments in the public healthcare network. It also provides access to the Digital National Vaccination Card, allowing Brazilians to check and verify all vaccines administered in both public and private networks, including the COVID-19 vaccine. ConecteSUS also offers a list of services and consultations available to the Brazilian population, including hospitals, health centers, emergency services, and popular pharmacies. It further lists maternity hospitals, health gyms, specialty centers, and psychosocial care centers available across the country. Additionally, it offers guidance on organ and blood donation. ConecteSUS is also developing a ‘superapp’, where more and more information will be available through the system, creating opportunities for startups to connect their projects and expand usage possibilities. These DataSUS records will be part of the National Health Data Network, which will be a large repository for information exchange, hosting both public and private data.
Another important topic discussed was the role of the patient as a central actor in the future. Aspects such as reducing wait times and enabling appointment scheduling are very practical to resolve, yield concrete results, and can reflect real improvements based on data. The pharmaceutical industry needs to evaluate everything under development and identify what may be of greatest interest, such as supporting efforts to organize clinical protocol data for specific lines of care. Certainly, appointment scheduling and reducing wait times are priorities for the government, and this could be a good opportunity for collaboration.
In the first quarter of 2023, the Digital Health Committee held a public meeting with the participation of various representatives from companies affiliated with Sindusfarma to address the topic of telemedicine in the context of the discussions surrounding the Ministry of Health’s Strategic Digital Health Plan.
Global Digital Health Strategy 2020–2025 WHO
From 2005 to 2020, WHO undertook significant efforts to develop a 5-year Demographic and Health Surveys (GDHS) through close consultation with member states and input from relevant stakeholders, identifying priority areas including those where WHO should focus its efforts. In this plan, digital health was defined as ‘the field of knowledge and practice associated with the development and use of digital technologies to improve health’, an expansion of the e-Health concept. The GDHS 2020–2025, developed through consultative processes, was ultimately endorsed by the 73rd World Health Assembly.
As outlined in the GDHS, digital health aims to focus on the patient by enabling better-informed health practices that would lead to improved healthcare and reduced costs, resulting from higher-quality health services enabled through the use of health-related technologies, including artificial intelligence (AI).
The GDHS acknowledges that 1) the institutionalization of digital health within national health systems requires decisions and commitments from countries, in accordance with their respective cultures, values, and available resources; 2) successful digital health initiatives require an integrated strategy among the many stakeholders in each country; 3) the appropriate use of digital health technologies requires protection against misinformation, fraud, and the improper use of health data; and 4) there is an urgent need to address the major barriers faced by less-developed countries in implementing digital health technologies.
The implementation of the GDHS is structured around four main strategic objectives: 1) to promote global collaboration and foster the transfer of knowledge about digital health; 2) to advance the implementation of country-specific digital health strategies; 3) to strengthen digital health governance at the global, regional, and national levels; and 4) to advocate for people-centered health systems enabled by digital health.
These objectives are further organized into short-, medium-, and long-term goals. According to the GDHS, each of these objectives would be achieved through actions to be undertaken by member states, the Secretariat (Technical and Administrative Staff of WHO), and partners (other agencies within the United Nations system, countries, and additional stakeholders, including the formation of new ones).
Interviews
The primary objective of the interviews was to understand, from diverse perspectives, the challenges of healthcare in Brazil, as well as the use and benefits of new digital technologies, particularly telemedicine, and identify critical partners and their roles in implementing the Digital Health Strategic Plan. Additional aims included understanding patient awareness regarding illness, prevention, and treatment; assessing the need for regulatory adaptation; and identifying key aspects of a sustainable health system.
The questions addressed nine important aspects to clarify the current healthcare scenario in Brazil and identify main challenges and opportunities from various viewpoints.
Below is an analysis of the different perspectives brought by the interviewees, highlighting converging and complementary points.
Health Challenges
All interviewees pointed to the lack of an integrated electronic and digital infrastructure system as a major challenge. This system could help address other reported issues, such as resource waste, absence of electronic health records, difficulty in communicating with remote healthcare units, and expenditure management. Two interviewees also noted the fragmentation in the health service delivery and insufficient federal funding for certain states and hospitals. A shortage of specialized professionals in remote regions was mentioned, linked to the lack of a policy for human resource development. The siloed operation of organizations and lack of coordination were also highlighted as major issues.
New Technologies
Interviewees mentioned digital platforms that enable telemedicine and patient monitoring, with two also referencing platforms for diagnostics and electronic prescriptions. To complement the above information, patient support programs offered by the pharmaceutical industry were brought up with concern, as many do not adequately meet patient needs. Robotic surgeries using metaverse technology were cited as rapidly evolving, particularly in the private sector, potentially expanding access to healthcare. Generative artificial intelligence (AI) (e.g. ChatGPT) was identified as a promising but challenging trend.
Telemedicine’s Potential Contribution to Increasing Access
All interviewees understand that the primary contribution of telemedicine is expanding access to healthcare for patients in remote regions. This will enable connections between general practitioners in these areas and specialists in major centers, improving patient care and contributing to the training of local medical professionals. From a patients’ association perspective, telemedicine facilitates faster service and accurate diagnoses, crucial especially for rare diseases. One concern raised was that certain specialties may or may not be suitable for telemedicine, warranting specific studies and regulations. Telemedicine may also allow for more efficient use of public funding by enabling more accurate diagnoses, frequent patient monitoring, and avoidance of unnecessary travel. In addition to that, teleconsultations, especially when paired with digital prescriptions, offer quicker access to treatment and allow for better monitoring of the patient journey (therapeutic adhesion, monitoring, digital acquisition, and dispensation).
Critical Stakeholders and Their Roles
Although the same critical stakeholders were cited by all, their roles varied according to each interviewee’s perspective. The former health secretary viewed the government as a leader; the patient association considered it a funder; Sindusfarma regarded it as a supporter of digital health. The pharmaceutical industry was unanimously identified as a critical actor, functioning as a supporter. The former secretary urged foreign-capital companies to play a more active role by sharing successful international digital health models and funding academic research to develop locally adaptable products. The patients’ association leader also called on the industry to disseminate accurate information on diseases, prevention, and treatment. Technology service providers were expected to ensure user security. Health professionals were deemed crucial by all; the patients’ association leader emphasized responsible use of telemedicine, avoiding practice outside one’s specialty, and ethical management of public resources. Sindusfarma’s president encouraged medical professionals to embrace technology. The CEO offered a strategic view, advocating for coordinated efforts among pharmaceutical associations, health plans, hospitals, and pharmacy networks to define common principles for health digitalization and telemedicine implementation.
Benefits to Patients
The main benefit attributed to telemedicine was improved access to information, diagnosis, specialists, treatment, and more precise monitoring, often without needing to travel. Other benefits regarding the evolution of digital health cited included the potential for electronic health records and prescriptions, delivery of medications, greater efficiency in exams by avoiding repetition, and increased access to health information. One interviewee noted that monitoring also supports treatment adherence and disease prevention.
Regulatory Environment
Interviewees generally found the regulatory environment favorable, noting the legislation that now regulates telehealth. However, more detailed regulations are needed on operational issues like payment models, teleconsultation rules in pharmacies, and specialties requiring initial in-person consultations. The patients’ association leader called for better regulation of industry-run patient support programs, which, in her view, often confuse patients and focus narrowly on enrollment and treatment continuation without providing useful information or broader support, which could be important for the patient and relatives. One interviewee stressed that full implementation of telehealth requires raising awareness, with cultural transformation now being the focus.
Definition of Sustainable Health
One interviewee defined sustainable health as the ability ‘to provide high-quality care to all, in a satisfactory manner, including access to innovative technologies without infrastructure or access barriers’. Others focused more on the ‘how’ to turn it into something more sustainable, including focusing on digital health (especially system integration), efficient resource allocation, sector collaboration with aligned guidelines, and ethical, law-compliant conduct.
Associations’ Contribution to Sustainability
Interviewees broadly agreed that associations should support digital health and all initiatives aimed at more efficient public resource allocation. The patients’ association also emphasized its vital role in empowering patients and families with accurate, timely information to ensure access to specialists, early diagnosis, and treatment. The digital solutions company highlighted the importance of coordination among sector associations to define common principles for health digitalization and telemedicine.
Results and Discussion
The results presented here are based on the data gathered during the participatory research phase, which allowed for thematizing key aspects observed and analyzed, contributing to the study’s objectives.
The most relevant themes identified were grouped into four spheres: digital, educational, legal, and political-economic.
Figure 1 lists these identified themes alongside possible strategic actions for the pharmaceutical industry and the Sindusfarma association, with a patient-centered focus. These actions aim to promote the full implementation of telemedicine, thereby: 1) expanding the number of patients aware of diseases, prevention, diagnosis, and treatments; 2) facilitating care by specialized professionals for proper diagnosis and treatment referral; 3) enabling patient monitoring, which supports treatment adherence and detection of adverse events, contributing to pharmacovigilance; and 4) promoting the use of electronic prescriptions for quicker access to treatments by patients.
Pharmaceutical companies may operate differently, rethinking their processes, ways of interacting with partners, and strategic plans, particularly their training programs, to upskill personnel in emerging areas. The study results highlight significant opportunities for pharmaceutical companies and for Sindusfarma, which represents the sector. The practical recommendations could help scale up telemedicine use, increasing patient access to disease awareness, diagnosis, and treatment.
The proposed actions for both the pharmaceutical industry and Sindusfarma are outlined in Appendix 1 and can be discussed internally by the respective companies and within the association’s working groups, committees, and subcommittees.
Final Considerations
This research offers a practical contribution to the pharmaceutical sector by presenting a map of opportunities for the expansion of telemedicine practices, with suggested actions for pharmaceutical industries and considerations for enhancing the positive agenda of Sindusfarma in the coming years, aiming to expand access to healthcare and position the entity as a strategic partner. These opportunities lie within the digital, legal, economic, and educational domains.
The implementation of these suggestions requires 1) professional resources within pharmaceutical companies with specific competencies and knowledge; 2) possibly, a review of internal processes and policies; 3) risk analysis; 4) revision of the strategic plan; 5) strengthening institutional relations, with participation in several committees and associations; and 6) expanding the network of relationships, especially in light of new stakeholders in the sector.
These opportunities suggest that industries should rethink how they create, deliver, and capture value. When building their institutional, commercial, and marketing strategies, they should consider the Strategic Digital Health Plan, particularly because it is a state plan and not a government plan. Intensifying disease prevention campaigns and stimulating discussions around the appropriate use of the healthcare system are also important commitments.
Patient support programs can become more comprehensive, incorporating diagnostic tests, critical follow-up exams, counseling for families and caregivers, nutritional and psychological support, the provision of electronic devices for disease monitoring, access to telemedicine platforms when necessary, among others. Hosting events to promote dialogue and provide educational support to healthcare professionals and patients (in digital literacy) can significantly contribute to the adoption of these technologies. A more active engagement by the industry with representative entities and associations is fundamental to foster debate and suggesting necessary legislative adaptations.
Moreover, reviewing how the industry interacts with all its partners, its communication channels, and the integration of those channels is undoubtedly necessary. Another identified opportunity relates to continuous improvement in internal processes and the training of professionals. The adaptation of products for delivery and use in remote areas is also relevant, as telemedicine has the potential to expand access not only to information but also to treatment.
In light of cooperation and partnership between the public and private sectors, the prioritization of the identified opportunities should be carried out by the entity in alignment with the Secretariat of Digital Health and Information at the Ministry of Health, considering its Strategic Digital Health Plan.21
The results of this research underscore the importance of the ecosystem and the need for coordination among its members’ strategies so that the healthcare system in Brazil can become sustainable.
It is essential that entities like Sindusfarma and associations such as Interfarma and others lead the debate among their members and position themselves as strategic partners in this digital transformation. The mentioned plan proposes to transform the relationships among the ecosystem’s members and meaningfully includes the technology sector, as well as other new participants.
The research also highlights the need for legislative adaptations in several relevant areas, such as AI, Privacy and Data Protection Law, Patient Support Programs, delivery of medications in remote areas using drones, Clinical Research, Pharmacovigilance, and regulation that allows pharmaceutical industry support for telemedicine (or even Telehealth), without obviously interfering in the freedom and independence of healthcare professionals in prescribing treatment.
Author Contributions
The author is responsible for all aspects of the development of this article.
Data Availability Statement (DAS), Data Sharing, Reproducibility, and Data Repositories
The author did not create a repository for the data used. The references direct the readers to the source of the materials.
Application of AI-Generated Text or Related Technology
None were used.
Acknowledgments
The author acknowledges the president of Sindusfarma and the leaders of the digital committee of Sindusfarma.
References
- Organização Pan-Americana Da Saúde/Organização Mundial Da Saúde (OPAS/OMS); Oliel S. OMS destaca 13 maiores desafios de saúde para a próxima década. Nações Unidas; 2020 [cited 2025 Mar 11]. Available from: https://news.un.org/pt/story/2020/01/1700342#:~:text=OMS%20destaca%2013%20maiores%20desafios%20de%20sa%C3%BAde%20para%20a%20pr%C3%B3xima%20d%C3%A9cada,-13%20Janeiro%202020&text=Ag%C3%AAncia%20da%20ONU%20destacou%20temas,%C3%A9%20poss%C3%ADvel%20faz%C3%AA%2Dlo.%E2%80%9D. Access on: March 17, 2023.
- Boni AA, Abremski D. Commercialization challenges and approaches for digital health transformation. J Commer Biotechnol. 2022;27(1). https://doi.org/10.5912/jcb1024
- Magretta J. Why business models matter. Harvard Business Review; 2002, p. 3–8.
- Osterwalder A, Pigneur Y, Tucci CL. Clarifying business models: Origins, present, and future of the concept. Commun Assoc Inf Syst. 2005;16(1):1. https://doi.org/10.17705/1CAIS.01601
- Casadesus-Masanell R, Ricart JE. From strategy to business models and onto tactics. Long Range Plann. 2010;43(2–3): 195–215. https://doi.org/10.1016/j.lrp.2010.01.004
- Zott C, Amit R, Massa L. The business model: Recent developments and future research. J Manag. 2011;37(4):1019–42. https://doi.org/10.1177/0149206311406265
- Brandenburger AM, Stuart HW, Jr. Value-based business strategy. J Econ Manag Strategy. 1996;5(1):5–24. https://doi.org/10.1111/j.1430-9134.1996.00005.x
- Lepak DP, Smith KG, Taylor MS. Value creation and value capture: A multilevel perspective. Acad Manag Rev. 2007;32(1):180–94. https://doi.org/10.5465/amr.2007.23464011
- Gassmann O, et al. Leading pharmaceutical innovation: How to win the life science race. Berlin: Springer, 2018.
- Saúde Digital News. Afya acelera expansão em serviços digitais médicos e anuncia aquisição da RX PRO. Saúde Digital News; October 1, 2021 [cited 2023 Mar 03]. Available from: https://saudedigitalnews.com.br/01/10/2021/afya-acelera-expansao-em-servicos-digitais-medicos-e-anuncia-aquisicao-da-rx-pro/
- Flash Courier. Flash Courier projeta dobrar número de entregas de produtos certificados pela Anvisa em abril. Flash Courier, São Paulo; 2020 [cited 2023 Mar 03]. Available from: https://flashcourier.com.br/blog/flash-courier-projeta-dobrar-numero-de-entregas-de-produtos-certificados-pela-an-5
- Alami H, Gagnon M-P, Fortin, J-P. Digital health and the challenge of health systems transformation. MHealth. 2017;3. https://doi.org/10.21037/mhealth.2017.07.02
- Sindusfarma. Estatuto Social Sindusfarma. Sindusfarma; 2019 [cited 2023 Mar 03]. Available from: https://sindusfarma.org.br/informativos/estatuto_social_sindusfarma.pdf
- Brasil. Você conhece o ConecteSUS? gov.br; 2021 [cited 2023 Mar 07]. Available from: https://www.gov.br/pt-br/noticias/saude-e-vigilancia-sanitaria/2021/04/voce-conhece-o-conecte-sus
- Brasil. Ministério da Saúde. A Estratégia Brasileira. gov.br; 2017 [cited 2023 May 16]. Available from: https://www.gov.br/saude/pt-br/assuntos/saude-digital/a-estrategia-brasileira/a-estrategia-brasileira
- Brasil. Ministério da Saúde. Estratégia de Saúde Digital para o Brasil 2020-2028. Brasília: Ministério da Saúde; 2020.
- Brasil. Decreto 11.358 de 1º janeiro de 2023. Aprova a Estrutura Regimental e o Quadro Demonstrativo dos Cargos em Comissão e das Funções de Confiança do Ministério da Saúde e remaneja cargos em comissão e funções de confiança. Diário Oficial da União; 2023.
- Universidade Aberta do Sistema Único de Saúde (UNA-SUS). Conheça as sete prioridades da Estratégia de Saúde Digital para o Brasil. UNA-SUS; 2023 [cited 2023 Mar 07]. Available from: https://www.unasus.gov.br/noticia/conheca-as-sete-prioridades-da-estrategia-de-saude-digital-para-o-brasil
- Sindusfarma. Comitês e grupos de trabalho. Sindusfarma; 2022 [cited 2023 Mar 07]. Available from: https://sindusfarma.org.br/sindusfarma/comites-e-grupos-de-trabalho#business-support
- Brasil DataSUS. Ministério da Saúde. [cited 2025 Jul 02]. Available from: https://datasus.saude.gov.br
- Sindusfarma. Strategic Plan 2023-2026 [cited 2025 Jul 02]. Available from: https://sindusfarma.org.br/en/sindusfarma/about/strategic-plan
Copyright Ownership: This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0. The authors of this article own the copyright.
Appendix 1
Suggested actions for the pharmaceutical industry and action proposals for the organization
| Identified opportunities | Suggestions for the pharmaceutical industry | Action proposals for the organization |
| Digital: Modernization of multicenter clinical research and use of AI |
|
Adapt legislation/regulation on research and the use of AI, to allow the use of AI systems for patient recruitment by cross-referencing medical records, reports, and data with study protocols. Besides recruitment, it is important to regulate the use of AI in data analysis, detection of adverse events, and treatment personalization based on genetic characteristics. |
| Educational: Patient as protagonist/Empowerment of patients and families |
|
|
| Digital: Data organization and management |
|
Position the industry as an active player, not just a supporter. |
| Educational: Human resource development policy – public servants and pharmaceutical industry professionals |
|
Partnership with academia and industry to promote scientific events and continuing education programs for professionals (telehealth/AI/clinical research – telemedicine). |
| Economic: Greater coordination among all sector organizations |
|
Union of efforts with AMCHAM, INTERFARMA, Patient Associations, Brazilian Medical Association, and other relevant associations. |
| Digital: Continuous improvement of pharmacovigilance quality to guide efficient use of medicines by doctors and patients |
|
Creation of a consortium for a pharmacovigilance monitoring center and for monitoring appropriate and personalized medicine use. |
| Digital: Continuous improvement of PSP offered by the pharmaceutical industry |
|
|
| Educational: Education of healthcare professionals in telemedicine (or more broadly, telehealth) |
|
|
| Economic: Products adapted for remote regions |
|
Regulation for drone use in product deliveries. Draft a white paper compiling international experience on the subject. |
| Legal: Support for the Digital Health Strategy |
|
Define common principles and guidelines for digital health modernization and the use of telemedicine (or telehealth) and its components. |
| Source: Elaborated by the author. AI: artificial Intelligence; AMCHAM: American Chamber of Commerce; ANVISA: Brazilian Health Regulatory Agency; PSP: Patient Support Program; Proadi-SUS: Support Program for the Institutional Development of the Unified Health System; SUS: Brazil Unified Health System. |
||
Addendum
Resumo
Este artigo apresenta uma análise acerca do impacto da telemedicina na indústria farmacêutica. Realizou-se uma pesquisa participante, com acompanhamento dos trabalhos do Comitê de Saúde Digital e análise dos trabalhos e discussões ocorridos de Marketing e Business Support, Regulatório, Estratégico Jurídico e Acesso, além de entrevistas com representantes do setor. Avaliou-se as Estratégias Global e Brasileira de Saúde Digital e o modelo de negócio da indústria farmacêutica para identificar o impacto da telemedicina no modelo de negócio. Apresentou-se um mapa de oportunidades para ampliação da prática de telemedicina, com sugestões de ações às indústrias farmacêuticas e considerações de projetos para o Sindusfarma, visando contribuir para o Plano Estratégico de Saúde Digital, para a inovação e modernização do setor e para a ampliação do acesso aos pacientes a informação, prevenção, diagnóstico e tratamento.
Resumen
Este artículo analiza el impacto de la telemedicina en la industria farmacéutica Comité de Salud Digital del Sindicato de la Industria de Productos Farmacéuticos del Estado de São Paulo (Sindusfarma). Se utilizó una investigación participante, junto a los trabajos del y análisis de los trabajos y debates ocurridos en sus comités de Marketing y Business Support, Reglamentación, Estrategia Jurídica y Acceso, y entrevistas con representantes del sector. Se evaluaron las Estrategias Globales y Brasileñas de Salud Digital y el modelo de negocio de la industria farmacéutica para identificar el impacto de la telemedicina en el modelo de negocio. Se presentó un mapa de oportunidades para la expansión de la práctica de la telemedicina; fueron propuestos acciones y proyectos, con vistas a contribuir al Plan Estratégico de Salud Digital, a la innovación y modernización del sector y ampliación del acceso de los pacientes a información, prevención, diagnóstico y tratamiento.