ORIGINAL RESEARCH
A Modern Informatics-Based System for Telemedicine: Adoption and Adherence in a Real-Life Study
Stefano Palazzo, MSc, P.Eng1,2,3  ; Cataldo Piarulli1,2; Marco Zurlo, MD4,5; Costantino Giuseppe Michele Della Corte, MD6; Silvio Tafuri, MD, PhD6; Marcello Albanesi, MD, PhD1,2,7,8
; Cataldo Piarulli1,2; Marco Zurlo, MD4,5; Costantino Giuseppe Michele Della Corte, MD6; Silvio Tafuri, MD, PhD6; Marcello Albanesi, MD, PhD1,2,7,8 
1“M. Albanesi” Allergy and Immunology Unit, Bari, Italy; 2The Allergist, Bari, Italy; 3Department of Engineering and Science, Universitas Mercatorum, Rome, Italy; 4Department of Medicine, University of Verona, Piazzale L.A. Scuro, Verona, Italy; 5Allergy Unit and Asthma Center, Verona Integrated University Hospital, Verona, Italy; 6Department of Biomedical Science and Human Oncology, Aldo Moro University of Bari, Bari, Italy; 7Allergolys, Paris, France; 8Department of Interdisciplinary Medicine, University of Bari Aldo Moro, Bari, Italy
Keywords: Adherence, allergology, control assessment test, digital health, digital questionnaires, healthcare, informatics, personalized medicine, RCAT, rhinitis, telemedicine, UAS, UAS7, Urticaria Activity Score
Abstract
Purpose: This study aims to develop an integrated telemedicine model to enhance the management and treatment of allergic disorders. Challenges like the completion of standardized clinical questionnaires, for example, the Urticaria Activity Score 7 (UAS7) for chronic urticaria and the Rhinitis Control Assessment Test (RCAT) for chronic rhinitis are also addressed. The goal is to improve data-collection methods, increase patient adherence and assist healthcare providers with timely and personalized clinical data interpretation.
Methods: The system was built on WordPress, and the interface was created using Python to generate electronic medical records. Each record, placed in the corporate cloud, contains certain questionnaires that are available to the patients via individual links. The data were processed using MATrix LABoratory (MATLAB) to evaluate adherence and outcomes.
Results: Among 100 patients, 76 patients reported completing their questionnaires via telemedicine, with 81% of the Urticaria Activity Score (UAS) being adherent compared to 57% for the RCAT. Adherence declined with the progression of the treatment cycles, showing a downtrend in patient input during prolonged therapies.
Conclusions: Telemedicine is useful to control clinical information in the field of allergology. On the other hand, the decrease in adherence with longer treatments calls for specific solutions, including reminders to patients. Subsequent development will aim to use artificial intelligence to broaden telemedicine for other allergic diseases, thus improving its precision and, above all, patient care.
Plain Language Summary
This study developed a simple, low-cost telemedicine system to help patients with allergic rhinitis and chronic urticaria track their symptoms remotely. Using available tools (a website and Excel forms), patients completed validated questionnaires (Urticaria Activity Score 7 [UAS7] for chronic urticaria and the Rhinitis Control Assessment Test [RCAT]) from home. The aim was to improve the monitoring of allergic diseases and assess how well patients adhere to digital follow-up methods.
Among 100 participants, 76% successfully completed their questionnaires online. Adherence was higher among patients with chronic urticaria (81%) than those with allergic rhinitis (57%). Patients who were younger or had higher education levels were slightly more likely to engage with the system, although age differences were not statistically significant.
These findings suggest that telemedicine can be an effective tool in allergy care when designed with simplicity and accessibility in mind. Future plans include developing a mobile app with reminders to further improve long-term adherence and expand digital monitoring to other allergic conditions.
Citation: Telehealth and Medicine Today © 2025, 10: 604
DOI: https://doi.org/10.30953/thmt.v10.604
Copyright: © 2025 The Authors. This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0. The authors of this article own the copyright.
Submitted: June 30, 2025; Accepted: August 29, 2025; Published: December 8, 2025
Corresponding Author: Stefano Palazzo, Email: stefano.palazzo@centroalbanesi.com
Competing interests and funding: The authors have no relevant financial or non-financial interests to disclose.
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
With the advance of personalized medicine, more sophisticated and targeted tools are essential to manage a patient’s allergic conditions. Telemedicine has a fundamental role in allergology to address the growing need for personalized medicine. Telemedicine, with its ability to provide healthcare services remotely, represents a unique opportunity to improve access to care and the management of allergic diseases.1,2
In clinical allergology and immunology, several standardized questionnaires are available to evaluate the impact of allergic, respiratory and skin diseases on quality of life, symptom control and therapy effectiveness.
Within this landscape, clinicians can choose among many validated, condition-specific questionnaires (including pediatric and abbreviated versions) for respiratory, food and skin allergies, supporting standardized evaluation of symptoms, disease control and quality of life.
The adherence rate to telemedicine varies significantly depending on the context. Moreira et al. highlighted the fact that adherence to telemedicine treatments can range from 15% to 70%. It was also observed that the use of diverse techniques and the implementation of shorter programs are associated with improved adherence.3 Furthermore, for respiratory allergic diseases like asthma, remote monitoring shows promise to improve symptom control and treatment adherence.4
UAS7 and RCAT
Chronic spontaneous urticaria and allergic rhinitis are among the most common clinical conditions in allergic patients. This study focuses on the use of the Urticaria Activity Score 7 (UAS7) and the Rhinitis Control Assessment Test (RCAT) in clinical allergology.
The UAS7 and RCAT are the most suitable questionnaires for the assessment of urticaria and rhinitis. They are important tools in research and in clinical practice, and above all, they are essential for each patient’s assessment of their diseases (being relatively simple, very specific, and providing reliable information for follow-up and treatment).1,5,6
The UAS is based on two main parameters: the number of wheals, scored from 0 (no wheals) to 3 (more than 50 wheals per day), and the intensity of itching, ranging from 0 (no itching) to 3 (severe itching). These two scores are added daily to obtain a maximum value of 6. A weekly version of the score, the UAS7, is also available, where “7” refers to the number of consecutive days for which the score is calculated. By summing the daily scores over 1 week, the UAS7 ranges from 0, indicating no symptoms, to 42, reflecting maximum disease activity.
The UAS is recommended by major international guidelines, such as those from the European Academy of Allergology and Clinical Immunology, Global Allergy and Asthma Excellence Network (GA2LEN), and World Allergy Organization (WAO). It is widely used in clinical practice and research to monitor disease progression and evaluate treatment efficacy. Despite its usefulness, it has limitations, such as excluding angioedema and relying on the patient’s subjective perception to assess itching intensity. Nevertheless, it remains a fundamental tool for the systematic monitoring of chronic spontaneous urticaria.
The RCAT questionnaire is designed to evaluate the control of allergic chronic rhinitis in patients.6 It contains six questions covering various aspects of chronic rhinitis, including the frequency of symptoms, the impact on daily activities, and the overall well-being of the patient. The RCAT is a valuable tool to identify patients who need treatment adjustments, thereby improving the management of allergic chronic rhinitis. Due to their ease of use, both questionnaires can be administered during clinical visits or via telemedicine, allowing for regular and timely monitoring of the patient’s condition.
Despite their proven usefulness, UAS and RCAT often present adherence difficulties for patients, especially when administered in paper format.7
Scientific studies highlight the fact that common challenges include forgetfulness, difficulty in understanding the questions, and the perception of the questionnaire as an overly complex process. To overcome these difficulties, the adoption of digital solutions and telemedicine is crucial.8
The scientific literature also reveals that creating a digital platform for administering the questionnaires allows patients to complete their responses easily and quickly using mobile devices or computers.9
Additionally, the integration with automatic reminder systems significantly improves adherence, ensuring that the questionnaires are completed within the expected timeframes.
Goals and Objectives
This study focuses on the creation of a system for managing and accessing patient information in a telemedicine context and helping to improve adherence issues.
The aim of this project, which involves the adoption of telemedicine, is to adapt the questionnaires to technological advancements, such as the transition from paper documentation to digital management. Therefore, it is essential to create a system that facilitates the comprehensive visualization of the patient group and streamlines the quantification of their clinical data.
Another aim is the development of an integrated system for managing and accessing patient information specifically designed for a telemedicine context. This system not only facilitates the administration and completion of clinical questionnaires by patients but also improves the accuracy and timeliness of the clinical data collected. Furthermore, the digitization of data management allows for a clearer and more comprehensive visualization of the patient group, enabling healthcare professionals to monitor and quantify patients’ clinical conditions more effectively.10,11
Finally, the project described in this study aims to integrate telemedicine into allergology practice, with the goal of improving patient adherence, the quality of the collected data and ultimately, therapeutic outcomes. The evolution towards a digital management system represents a crucial opportunity for modern allergology, enabling it to address the current and future challenges of personalized medicine.
Materials and Methods
Patient Treatment Schemes and Follow-Up
A total of 100 patients were enrolled in this real-life study with a mean age of 32.95 ± 10.85 years (Figure 1A), including 49 males and 51 females (Figure 1B).
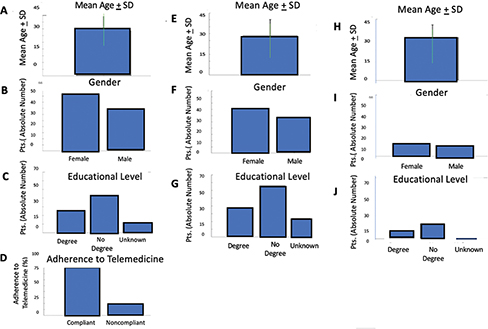
Fig. 1. Demographic characteristics, education level, and telemedicine adherence in the total study population. Mean age and standard deviation of the entire patient cohort (A) Numerical distribution of the entire cohort based on sex (B); Distribution of the entire cohort based on education level: graduates, non-graduates, and patients with undocumented education levels (C); Percentage of patients who adhered to telemedicine, calculated regarding the total patient cohort based on questionnaire responses (D); Mean age and standard deviation of all patients who adhered to telemedicine (E); Numerical distribution of all patients adhering to telemedicine based on sex (F); Distribution of patients adhering to telemedicine based on education level: graduates, non-graduates, and patients with undocumented education levels (G); Mean age and standard deviation of all patients who did not adhere to telemedicine (H); Numerical distribution of non-adherent patients based on sex (I); Distribution of non-adherent patients based on education level: graduates, non-graduates, and patients with undocumented education levels (L).
All the patients were treated according to the international guidelines for chronic urticaria and allergic rhinitis. The clinical features of the entire patient cohort, including diagnosis (chronic urticaria or chronic rhinitis), telemedicine adherence rate, age distribution, sex, and prescribed therapy, are shown in Appendix A, which provides a comprehensive overview of baseline characteristics for both diagnostic groups, allowing direct comparison between adherent and non-adherent patients.
In particular, 79 patients with chronic rhinitis and 21 patients with urticaria were analyzed. Patients with chronic rhinitis received the RCAT questionnaire, while patients with chronic urticaria received the UAS7 questionnaire.
In this study, the term “telemedicine” refers exclusively to the remote collection of clinical data through digital self-administered questionnaires (UAS7 and RCAT), without involving other forms of real-time clinical interaction.
The electronic version of both questionnaires was provided to each patient. Patients who explicitly requested the paper questionnaire were excluded from the study (digital-first enrollment; only patients consenting to electronic completion [UAS7/RCAT] were included).
No specific evaluation of patients’ ability or preference to use digital tools (i.e., no pre-screening for digital skills or device access) was conducted.
For all patients, information regarding education level was collected, including bachelor’s degree, master’s degree, and PhD qualifications.
All the patients received therapies according to the international guidelines for the standard treatments for allergic rhinitis and chronic urticaria. A monthly follow-up for the clinical efficacy of the treatment was performed.
This observational real-life study, conducted without experimental interventions or deviations from standard clinical practice, did not require Ethics Committee approval according to Italian regulations (Ministerial Decree 30/11/2021, implementing the (European) EU Regulation 536/2014), as it involved anonymized clinical data collected during routine care and complied with the Declaration of Helsinki and General Data Protection Regulation (GDPR) (EU 2016/679). All patients provided written informed consent for the processing of personal and health-related data before the clinical visit. All procedures performed were consistent with the standard of care.
Creation of Digital Medical Record
The system was implemented on a dedicated website, using the WordPress CMS and the HTML programming language. The front-end part of the website has a section dedicated to entering patient data (name and surname).
To enable automatic data entry into the back-end part of the website, a graphical interface was developed using Python. Specifically, the Tkinter library was used to create an efficient and intuitive Graphical User Interface (GUI), facilitating the physician’s interaction with the system by simplifying the process of creating, for each patient, a medical record containing the questionnaire related to the diagnosed pathology.
A technical description of the system’s functionality and the implemented solutions for managing digital medical records are provided in the Appendix B (“Technical Description of the Digital Medical Record Management System”).
Essential Components for Computerized Management
The programmer implemented the following:
- Python: Programming language, known for its versatility and accessibility, was used to design a free and intuitive system that locally creates a medical record for each patient on a corporate cloud storage service.
- Corporate cloud storage service was used as a secure and free data storage platform; it offers a practical and easily accessible solution for managing medical information. In this study, all clinical data were stored on Google Drive, chosen as a secure and reliable corporate cloud service. Google Drive ensures encryption at rest (AES-256) and encryption in transit (TLS/SSL), protecting data both during storage and transfer. Access to the Drive was restricted to authorized personnel only, with account-level security managed through Google’s Identity and Access Management system (two-factor authentication and activity logging were enabled to track access events). Patient questionnaires were distributed via individualized, revocable HTTPS links, avoiding open directories. Furthermore, predefined data retention periods and protected backups were enforced in accordance with GDPR principles of data minimization and integrity. This solution provided a safe, GDPR-compliant environment for long-term storage of anonymized clinical data, guaranteeing confidentiality, integrity, and availability as required by GDPR Article 32.12–14
From the patients’ perspective, the process of completing the questionnaires consists of two simple steps. The tools they see and use are:
-Website (www.marcelloalbanesi.com): In the dedicated section, the patient simply needs to enter their first and last name to access their questionnaire file, making the access process extremely intuitive.
-Excel File: Easy to fill out and widely compatible, as many PCs come with the Office suite, and many modern smartphones have pre-installed or easily downloadable apps that open Office files, including Excel files. This choice makes the user interface immediately familiar and simple to use.
Data Analysis
In addition, at the same time, an Excel archiving file was developed for data analysis, which includes detailed information for each patient, such as name, surname, age, sex, pathology, and the specific choice to adopt telemedicine.
Subsequently, data analysis was conducted entirely using MATLAB (MathWorks).
Language Editing and Translation Support
ChatGPT 4.0 (OpenAI) was used only to support the translation of specific words and technical terms from Italian to English and for minor language adjustments. The authors drafted the manuscript; no scientific content was generated by artificial intelligence (AI), whose role was strictly limited to linguistic assistance. Scientific literature acknowledges ChatGPT as a valuable tool for non-native authors in improving academic texts, enhancing international scientific communication.15–19 AI-assisted editing was reviewed and validated by a domain expert to ensure accuracy. As noted by Palazzo et al., the optimal approach combines human expertise with AI capabilities.20
Results
Initial Therapy
The study sample consisted of 100 patients with a mean age of 32.95 ± 10.85 years (Figure 1A, indicating a relatively young adult cohort with moderate variability), including 49 males and 51 females (Figure 1B, showing an almost perfectly balanced sex ratio).
Among all the patients, 32 had at least a university degree, 53 did not hold a degree, while for 15 patients there was no information available regarding their educational level (Figure 1C, non-graduates represent slightly more than half of the cohort, followed by graduates and a smaller fraction with unknown education).
Overall, adherence to telemedicine was 76% among patients who completed their questionnaires online, compared to 24% of patients who did not complete the digital questionnaires (Figure 1D, a marked preference and completion rate for the digital format).
Therefore, out of the 100 patients:
-76% (mean age: 29.47 ± 12.20 years, Figure 1E showing a lower mean age than the total sample: 38 males and 38 females, Figure 1F indicating perfect gender parity among adherents) successfully used the digital modality (telemedicine) for data compilation. Among these patients, 25 have at least one degree, 38 do not have a degree, and the educational level of 13 patients is unknown (Figure 1G, where non-graduates are the most represented group, followed by graduates, with a notable fraction of missing data).
-24% (mean age: 34.45 ± 10.06 years, Figure 1H with a higher mean age compared to adherents; 11 males and 13 females, Figure 1I showing a mild female predominance) did not complete the digital modality. The patients, after initially indicating their intention to complete the digital version and thus not requesting the paper questionnaire, ultimately did not complete it. Among these patients, 7 had at least one degree, 15 did not have a degree, and the educational level of 2 patients is unknown (Figure 1L, where non-graduates also predominate, followed by graduates, with minimal missing data).
Furthermore, in addition to the descriptive analysis, statistical tests were performed to assess whether the differences between compliant and non-compliant patients were significant.
Specifically, the t-test revealed no statistically significant difference in age between compliant and non-compliant telemedicine patients (P = 0.094640).
In summary, Figure 1 shows that telemedicine adherence in the overall cohort was high and did not appear to be strongly influenced by sex or educational level, while adherent patients tended to be slightly younger on average than non-adherent ones.
Chronic Urticaria – UAS7
As for chronic urticaria, 79% of all patients (mean age: 33.58 ± 10.84 years, Figure 2A, similar age to the total sample; 34 males and 45 females, Figure 2B indicating a modest female predominance) completed the UAS questionnaire.
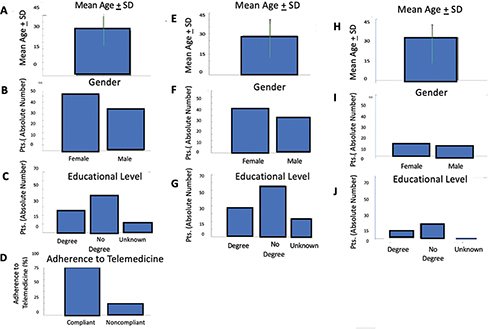
Fig. 2. Demographics, education level, and telemedicine adherence among patients with chronic urticaria (UAS7): Mean age and standard deviation of patients with chronic urticaria (A); Numerical distribution of the patient cohort with urticaria based on sex (B); Distribution of the patient cohort with urticaria based on education level: graduates, non-graduates, and patients with undocumented education levels (C); Percentage of patients who adhered to telemedicine, calculated regarding the total number of patients with urticaria, based on questionnaire responses (D); Mean age and standard deviation of all patients with urticaria who adhered to telemedicine (E); Numerical distribution of patients with urticaria adhering to telemedicine based on sex (F); Distribution of patients with urticaria adhering to telemedicine based on education level: graduates, non-graduates, and patients with undocumented education levels (G); Mean age and standard deviation of patients with urticaria who did not adhere to telemedicine (H); Numerical distribution of non-adherent patients with urticaria based on sex (I); Distribution of non-adherent patients with urticaria based on education level: graduates, non-graduates, and patients with undocumented education levels (L).
Among all patients with urticaria (regardless of adherence to telemedicine), 24 (30.38%) hold at least a university degree, 43 (54.43%) did not have a degree, while for 12 (15.19%) patients, no information regarding their education was provided (Figure 2C, with non-graduates forming the largest category).
In these patients with urticaria, adherence to telemedicine was found to be 81% among those who opted to use the digital mode for completing the questionnaires, compared to 19% of patients who did not complete the digital mode (Figure 2D, indicating a high adherence rate in this subgroup).
Specifically, out of the 79 patients:
-81% (mean age: 28.53 ± 11.69 years, Figure 2E showing a notably younger mean age than the non-adherent subgroup; 29 males and 35 females; Figure 2F confirming the female predominance) completed the form correctly. Among these patients, 19 (29.69%) had at least one degree, 33 (51.56%) did not have a degree, and the educational level of 12 patients (18.75%) was unknown (Figure 2G, where non-graduates clearly predominate, followed by graduates, with a substantial fraction unknown).
-19% (mean age: 35.26 ± 10.12 years, Figure 2H, higher than adherents; 5 males and 10 females; Figure 2I, with a stronger female predominance) did not complete the form. Some patients, after initially indicating their intention to complete the digital version and thus not requesting the paper questionnaire, ultimately did not complete it. Among these patients, 5 (33.33%) had at least one degree, and 10 (66.67%) did not have a degree (Figure 2L, with no missing data and a clear majority of non-graduates).
In summary, Figure 2 shows that patients with chronic urticaria demonstrated a very high adherence to telemedicine, particularly among younger individuals, with female patients slightly more represented.
Education level distribution was similar between adherent and non-adherent groups.
Rhinitis – RCAT Score
Twenty-one percent of total patients (mean age: 30.56 ± 11.23 years, Figure 3A, slightly younger than the total sample; 15 males and 6 females; Figure 3B indicating a marked male predominance) received the RCAT questionnaire.
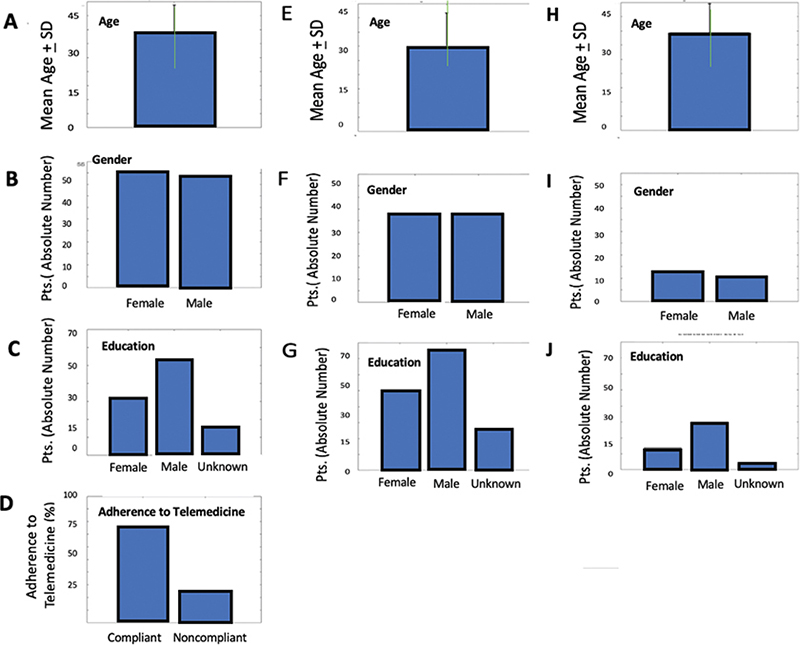
Fig. 3. Demographics, education level, and telemedicine adherence among patients with chronic rhinitis (Rhinitis Control Assessment Test [RCAT]): Mean age and standard deviation of patients with chronic rhinitis (A); Numerical distribution of the patient cohort with rhinitis based on sex (B); Distribution of the patient cohort with rhinitis based on education level: graduates, non-graduates, and patients with undocumented education levels (C); Percentage of patients who adhered to telemedicine, calculated regarding the total number of patients with rhinitis, based on questionnaire responses (D); Mean age and standard deviation of all patients with rhinitis who adhered to telemedicine (E); Numerical distribution of patients with rhinitis adhering to telemedicine based on sex (F); Distribution of patients with rhinitis adhering to telemedicine based on education level: graduates, non-graduates, and patients with undocumented education levels (G); Mean age and standard deviation of patients with rhinitis who did not adhere to telemedicine (H); Numerical distribution of non-adherent patients with rhinitis based on sex (I); Distribution of non-adherent patients with rhinitis based on education level: graduates, non-graduates, and patients with undocumented education levels (L).
Among all patients with rhinitis (regardless of adherence to telemedicine), 8 (38.09%) hold at least a university degree, 10 (47.62%) did not have a degree, while for 3 (14.29%) patients, no information regarding their education level was provided (Figure 3C, showing a relatively balanced graduate/non-graduate split with a small unknown group).In these patients with rhinitis, adherence to telemedicine was found to be 57% among those who opted to use the digital mode for completing the questionnaires, compared to 43% of patients who did not complete the digital mode (Figure 3D, revealing a notably lower adherence compared to urticaria patients).
Specifically, out of the 21 patients:
-57% (mean age: 29.87 ± 8.91 years, Figure 3E, comparable to the non-adherent; subgroup nine males and three females, Figure 3F reflecting a strong male predominance) completed the form. Among these patients, six had at least one degree, five did not have a degree, and the educational level of one patient is unknown (Figure 3G, with graduates slightly more represented).
-43% (mean age: 31.25 ± 13.77 years, Figure 3H, marginally higher than adherents; six males and three females, Figure 3I maintaining male predominance) did not complete the form. Some patients, after initially indicating their intention to complete the digital version and thus not requesting the paper questionnaire, ultimately did not complete it. Among these patients, two had at least one degree, five did not have a degree, and the educational level of two patients is unknown (Figure 3L, where non-graduates form the majority, with a relevant share of missing data).
In summary, Figure 3 shows that adherence among chronic rhinitis patients was markedly lower than in urticaria patients, with similar mean ages between adherent and non-adherent groups. A predominance of male patients was observed in both adherent and non-adherent subgroups.
In case of prolonged therapy requiring a second and third completion of clinical diaries, the details regarding patient adherence trends and the specifics of the administered questionnaires are provided in supplementary material Appendix C (“Adherence trends in prolonged telemedicine therapy for chronic urticaria and chronic rhinitis”).
Integrated Comparison of UAS7 and RCAT Cohorts
To provide a more comprehensive view of the results, in addition to the detailed descriptions provided in the previous sections, a comparative summary integrating the main demographic variables and adherence data has been prepared in Table 1.
This summary allows for immediate visibility of the differences between the two clinical groups considered (patients with chronic urticaria and patients with chronic rhinitis), highlighting both the different demographic profiles and trends in adherence behavior.
This makes the analysis more compact and easily interpretable, strengthening the overall coherence of the presentation and supporting the observations emerging from the detailed descriptive analyses.
Furthermore, building on this comparative summary, the complete set of within- and between-cohort statistical tests by age, sex, and education is reported in the Appendix D (“Associations Between Telemedicine Adherence and Demographic/Educational Characteristics”).
Discussion
Low-Cost Information Systems for Telemedicine
Highly simple, widely known, and user-friendly IT tools were employed, ensuring a cost-effective and efficient approach.
The adoption of such technologies not only meets the need to modernize the process of collecting and managing clinical data but also represents a fundamental step towards more personalized and patient-centered medicine.9,21
The use of simple, free, and open-access systems for telemedicine reduces costs and technical barriers and promotes digital inclusion, allowing more people to benefit from healthcare innovations. This approach democratizes access to care, making it more accessible and sustainable in the long term.
The significance of this development lies in having created an efficient telemedicine system using simple and low-cost IT tools, demonstrating that advanced technological solutions can be achieved even with limited resources, without compromising the quality of the service provided.22
The ability to collect real-time data and access clinical information allows for more timely and personalized interventions, thereby improving clinical outcomes and the quality of life for patients with allergies.
Patient Adherence to Telemedicine
In scientific literature, adherence rates to telemedicine show significant variability.
For instance, the study by Radojević et al. on patients with Parkinson’s disease highlighted the usefulness of the Adherence to Refills and Medications Scale (ARMS) in evaluating adherence levels in individuals with chronic conditions.23
Similarly, the study by Jongen et al. analyzed adherence to digital questionnaires in patients with multiple sclerosis. After 2 years, 56.8% of patients completed the low-frequency questionnaires, while adherence was significantly higher for questionnaires administered less frequently.40
On the other hand, Moreira et al. found that adherence to telemedicine can range between 15% and 70%. They also highlighted the fact that excessively lengthy interventions tend to reduce the effectiveness of telemedicine.3 Mosnaim et al. also highlighted the fact that the effectiveness of telemonitoring can be compromised by interventions that are too lengthy, with adherence rates that are variable and often reduced in these cases.4
In our case, consistent with the literature, higher adherence was observed for questionnaires administered at higher frequencies. Both questionnaires we employed were characterized by their user-friendly design and simplicity, which facilitated user participation.
The results of this study show that telemedicine can be used in allergology, with an adherence rate of 76%, indicating a majority of participants and demonstrating the potential for widespread acceptance of such systems.
This system also improves the management of patient information and facilitates doctor–patient communication in real time.
Interestingly, the number of patients who received the telemedicine system for the UAS7 card for the first time is higher than the number of patients who received the RCAT.
The increased adherence to UAS7 could be attributed to:
-the need for daily monitoring of symptoms (less likely to forget), as patients are driven by the need to resolve a sudden problem that arises from a suddenly arisen clinical condition;
-alteration of the quality of life of the patient with urticaria, which leads to greater commitment and adherence to the proposed therapeutic schemes, unlike patients suffering from asthma or chronic rhinitis who may perceive less urgency in seeking a solution, being now familiar with the persistence of their symptoms over time.
Furthermore, it was important to note that a high portion of patients (76 out of a total of 100) are already predisposed to use telemedicine solutions and recognize the benefits of more timely diagnosis and treatment. In this context, it is important to note that the study was based on consecutive enrollment, including only patients who accepted the electronic questionnaire format. The electronic format was the standard, and the paper questionnaire was available only upon explicit request, so no paper-based data were collected, and digital-vs-paper adherence could not be assessed.
Restricting inclusion to patients accepting electronic questionnaires introduces a self-selection bias (digital literacy/technology access/preferences) that may overestimate adherence. Therefore, its magnitude cannot be determined in the absence of a concurrent paper or mixed-mode comparator.
The goal is to adopt a modern, digital-first approach, in line with telemedicine trends and optimized for patient engagement and data reliability.
Since some patients choose, a priori, not to embrace telemedicine (opting for paper-based methods), they would have made the same choice regardless of the telemedicine system we implemented and how user-friendly it was.
Prior work suggests that electronic diaries can show consistently higher adherence than paper due to timestamps, reminders, and reduced backfilling, whereas in broader survey contexts, paper or mixed-mode can yield higher response than web-only. Overall, adherence is strongly influenced by key design features (reminders/UX) rather than mode alone.24–29
In addition, statistical analysis indicated that adherence was significantly influenced by the level of education, with graduates, non-graduates, and even patients with unknown education showing higher adherence rates compared to their non-adherent counterparts (P < 0.01). By contrast, no significant differences were observed in terms of age between adherent and non-adherent groups (P = 0.0946). These findings suggest that socio-educational background may play a more relevant role than age in determining the acceptance of telemedicine tools.
The implementation of this system demonstrated the potential of telemedicine in improving the management of patient information and facilitating communication between doctor and patient in real time.
Behavioral Determinants of Telemedicine Adherence
These adherence patterns can also be interpreted through established behavioral frameworks in digital health.
According to the “Technology Acceptance Model,” user engagement with a digital system is primarily driven by perceived usefulness and perceived ease of use. In this study, the UAS7 group, which showed higher adherence, may have perceived stronger immediate clinical benefits from frequent monitoring, thus reinforcing the perceived usefulness of the platform. Conversely, patients completing the RCAT (often managing long-standing and less acutely distressing symptoms) may have perceived lower short-term benefits, which could partially explain the reduced adherence.
Similarly, the “Health Belief Model” emphasizes the role of perceived severity and perceived susceptibility in shaping health-related behaviors. The higher adherence in urticaria patients could reflect a greater perceived severity and an increased motivation to engage in daily monitoring, while rhinitis patients may perceive their condition as less urgent, reducing motivation for repeated questionnaire completion.30,31
These interpretations highlight that interventions addressing perceived benefit, threat, and ease of use can improve engagement over time.
Study Limitations
This observational, digital-first study without a paper-based comparator, with differing sample sizes between UAS7 and RCAT and cycle-based follow-up, may limit generalizability and the power of some analyses, while retaining high pragmatic relevance. Moreover, the sample is not very large, so the findings should be confirmed in larger cohorts.
Future Development
Considering the results obtained, future developments of the telemedicine project in allergology could focus on several key areas. Firstly, it is possible to explore the integration of AI algorithms to analyze the collected data and provide decision support to doctors. AI could help identify patterns in patients’ symptoms and suggest personalized interventions based on historical and current data.
A timeline of key milestones (Figure 4) situates this proposal within the broader evolution of digital data collection in allergology – from early healthcare digitalization and formalized recordkeeping, through the emergence of standardized patient-reported outcomes, to our 2024 remote diary deployment.32–35
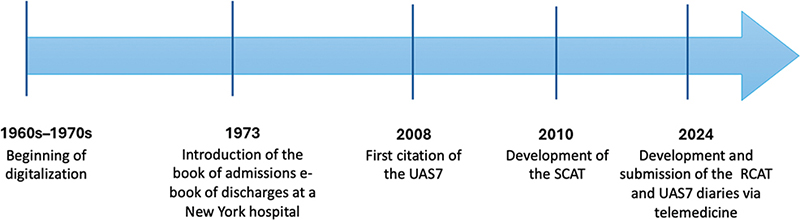
Fig. 4. Timeline of key milestones in digital data capture for allergology (1960s–2024). Milestones span early hospital digitalization (1960s–1970s)1,32 formal recordkeeping (1973),2,33 standardization of allergy PROs (UAS7, 20085,34 RCAT, 20106,35y and our 2024 telemedicine deployment for remote UAS7/RCAT diaries – providing the rationale for a next step via a mobile app with push notifications to sustain adherence over time. RCAT: Rhinitis Control Assessment Test; UAS7: Urticaria Activity Score 7.
Against this backdrop, developing a mobile application with push notifications represents the next logical step to address the adherence decay over time observed in our cohort by enabling timely reminders, real-time entry, and improved data completeness.
Another area of development involves expanding the platform to include additional questionnaires and monitoring tools for other allergic conditions, such as atopic eczema and food allergies. This would allow for more comprehensive and integrated support for patients with different allergic diseases. Moreover, it would be useful to develop a dedicated mobile application, which could further enhance accessibility and patient adherence, offering push notifications, personalized reminders, and a notification system that acts as a reminder to complete the digital questionnaires.36,37,38
The integration of a notification system represents an effective strategy to counteract forgetfulness, one of the main obstacles to patient adherence in using digital platforms for managing allergic conditions. Push notifications could be personalized based on the specific needs of each patient, sending reminders at optimal times of the day to minimize disruption of their daily activities.
In addition to the simple reminder function, the notification system could include motivational and informational messages to encourage patients to adhere to monitoring their condition. These messages could explain the importance of regularly completing the questionnaires for effective management of their allergy and provide tips on how to better manage their symptoms. Therefore, the notifications would remind patients to complete the questionnaires and encourage them to take an active role in managing their health.
Furthermore, the notification system could be integrated with questionnaire completion tracking features, sending alerts to both patients and doctors in case of non-completion. This would allow doctors to promptly intervene to resolve any adherence issues, ensuring continuous and accurate monitoring of the patient’s condition.
Built on these ideas and motivated by the historical progression summarized in Figure 4, a future roadmap could include the development of a prototype mobile application integrating the current digital system with advanced notification features and user-friendly interfaces. A pilot study could be planned to assess the feasibility and clinical utility of the system, focusing on user adherence, satisfaction, and usability metrics. This preliminary study would involve a small patient cohort and could serve to identify potential barriers, optimize interface design, and refine notification timing and content. The insights gained might guide the full-scale deployment of the platform and the integration of additional intelligent decision-support tools.
Contributors
Eng. Palazzo designed and implemented the computational code, performed data analysis, and drafted the manuscript. Mr. Piarulli assisted in patient data collection. Dr. Zurlo contributed to the literature review and assisted with English language editing of the manuscript. Dr. Della Corte curated and structured the therapy table. Dr. Tafuri provided ethical oversight and supervision regarding compliance with ethical guidelines. Dr. Albanesi conceived and supervised the study, secured financial support, and revised the manuscript.
Data Availability Statement (DAS), Data Sharing, Reproducibility, and Data Repositories
The data supporting the findings of this study are available from the corresponding author upon reasonable request. However, all relevant information, including the complete dataset structure, variables, and extensive analyses, are fully reported and discussed within the article. As such, external access to the raw data is not required for the interpretation, replication, or evaluation of the results presented.
Application of AI-Generated Text or Related Technology
Information regarding the use of AI-based tools for language editing and translation is provided in the Materials and Methods section, under the paragraph titled “Language editing and translation support”.
Ethics Approval Declaration
The study was conducted in a real-life observational context, without any experimental interventions, randomization, or modifications to standard clinical practice.
In accordance with Italian national regulations and the Declaration of Helsinki of 1964 and its subsequent amendments, observational non-interventional studies based on routine treatments and without active clinical experimentation do not require prior approval from an Ethics Committee nor registration in a clinical trial registry.39,40,41
Specifically, under current Italian law (Ministerial Decree of November 30, 2021, implementing EU Regulation 536/2014), observational non-interventional studies that use clinical data collected during routine care and do not involve therapeutic or diagnostic changes are not subject to Ethics Committee authorization or mandatory registration in public clinical trial databases.
The clinical data used had been previously collected for diagnostic and therapeutic purposes and were subsequently analyzed in aggregated form, without the publication of individual data and without any interference with the behavior or treatment of participants. Moreover, the questionnaires did not contain sensitive identifiers such as date of birth or full personal details, but only patients’ initials, further minimizing privacy risks. All individuals involved provided written informed consent for the use of their personal and clinical data in accordance with applicable privacy regulations.
Data collection, processing, and storage were carried out in full compliance with the General Data Protection Regulation (GDPR – EU Regulation 2016/679), ensuring appropriate safeguards for privacy and data security.
References
- Lang DM. The impact of telemedicine as a disruptive innovation on allergy and immunology practice. Ann Allergy Asthma Immunol. 2022;128(2):146–51. https://doi.org/10.1016/j.anai.2021.11.003
- Portnoy JM, Pandya A, Waller M, Elliott T. Telemedicine and emerging technologies for health care in allergy/immunology. J Allergy Clin Immunol. 2020;145(2):445–54. https://doi.org/10.1016/j.jaci.2019.12.903
- Moreira TC, Signor L, Figueiró LR, Fernandes S, Bortolon CB, Benchaya MC, et al. Non-adherence to telemedicine interventions for drug users: systematic review. Rev Saude Publica. 2014;48(3):521–31. https://doi.org/10.1590/S0034-8910.2014048005130
- Mosnaim G, Carrasquel M, Ewing T, Berty A, Snedden M. Remote monitoring in asthma: a systematic review. Eur Respir Rev. 2025;34(175):240143. https://doi.org/10.1183/16000617.0143-2024
- Kolkhir P, Giménez-Arnau AM, Kulthanan K, Peter J, Metz M, Maurer M. Urticaria. Nat Rev Dis Primers [Internet]. 2022 [cited 2025 Sep 9];8(1):61. Available from: https://www.nature.com/articles/s41572-022-00389-z
- Del Cuvillo A, Sastre J, Colás C, Navarro AM, Mullol J, Valero A. Adaptation to Spanish and validation of the rhinitis control assessment test questionnaire. J Investig Allergol Clin Immunol. 2020;30(3):175–81. https://doi.org/10.18176/jiaci.0420
- Liu W, Zeng Q, He C, Chen R, Tang Y, Yan S, et al. Compliance, efficacy, and safety of subcutaneous and sublingual immunotherapy in children with allergic rhinitis. Pediatr Allergy Immunol. 2021;32(1):86–91. https://doi.org/10.1111/pai.13332
- Beheshti L, Kalankesh LR, Doshmangir L, Farahbakhsh M. Telehealth in primary health care: a scoping review of the literature. Perspect Health Inf Manag. 2022;19(1):1n.
- Barbosa W, Zhou K, Waddell E, Myers T, Dorsey ER. Improving access to care: telemedicine across medical domains. Ann Rev Public Health. 2021;42:463–81. https://doi.org/10.1146/annurev-publhealth-090519-093711
- Kvedarienė V, Burzdikaitė P, Česnavičiūtė I. mHealth and telemedicine utility in the monitoring of allergic diseases. Front Allergy. 2022;3:919746. https://doi.org/10.3389/falgy.2022.919746
- Persaud YK. Using telemedicine to care for the asthma patient. Curr Allergy Asthma Rep. 2022;22(4):43–52. https://doi.org/10.1007/s11882-022-01030-5
- Masyhur Z, Rizaldy A, Kartini P. Studi Literatur Keamanan dan Privasi Data Sistem Cloud Computing Pada Platform Google Drive. shift [Internet]. 2021 [cited 2025 Sep 8];1(2):31–8. Available from: http://shift.sin.fst.uin-alauddin.ac.id/index.php/shift/article/view/15
- Mardi Y, Oktavia D. Socialization of the utilization of Google drive as an online storage medium for health workers in health facilities. ABDIMAS. 2025;8(1):338–46. https://doi.org/10.35568/abdimas.v8i1.5667
- Claybaugh CC, Chen L, Haried P, Zhou D. Risk and information disclosure in Google drive sharing of tax data. In: Nah FFH, Xiao BS, editors. HCI in business, government, and organizations [Internet]. Cham: Springer International Publishing; 2018, pp. 42–50 [cited 2025 Sep 8]. (Lecture Notes in Computer Science; vol. 10923). Available from: https://link.springer.com/10.1007/978-3-319-91716-0_4
- Wang Y. Reviewing the usage of ChatGPT on L2 students’ English academic writing learning. J Educ Human Soc Sci. 2024;30:173–8. https://doi.org/10.54097/dvjkj706
- Translating the Untranslatable: DeepL and ChatGPT on Academic Idioms | Linguistik, Terjemahan, Sastra (LINGTERSA) [Internet]. [cited 2025 Sep 8]. Available from: https://talenta.usu.ac.id/lingtersa/article/view/15086
- Banimelhem O, Amayreh W. Is ChatGPT a good English to Arabic machine translation tool? 2023 14th International Conference on Information and Communication Systems (ICICS) [Internet]. 2023 [cited 2025 Sep 9]; 1–6. Available from: https://ieeexplore.ieee.org/document/10330525/
- Chou W, Chow JC. Enhancing English abstract quality for non-English speaking authors using ChatGPT: a comparative study of Taiwan, Japan, China, and South Korea with slope graphs. Medicine (Baltimore). 2024;103(40):e39796. https://doi.org/10.1097/MD.0000000000039796
- Afifah. Using artificial intelligence (AI) for academic writing: How not to cross the line [Internet]. International Society for Technology, Education, and Science; 2024 [cited 2025 Sep 8]. Available from: https://eric.ed.gov/?id=ED673161
- Palazzo S, Chaoul N, Albanesi M. Human vs machine in bioengineering allergology: a comparative analysis of conventional vs innovative methods for quantifying allergological skin prick tests. 2024 [cited 2025 Sep 9]. Available from: https://www.unimercatorum.iris.cineca.it/handle/20.500.12606/24318
- Smolinska S, Popescu FD, Izquierdo E, Antolín-Amérigo D, Price OJ, Alvarez-Perea A, et al. Telemedicine with special focus on allergic diseases and asthma-Status 2022: an EAACI position paper. Allergy. 2024;79(4):777–92. https://doi.org/10.1111/all.15964
- Dramburg S, Walter U, Becker S, Casper I, Röseler S, Schareina A, et al. Telemedicine in allergology: practical aspects: a position paper of the Association of German Allergists (AeDA). Allergo J Int. 2021;30(4):119–29. https://doi.org/10.1007/s40629-021-00167-5
- Radojević B, Dragašević-Mišković NT, Milovanović A, Svetel M, Petrović I, Pešić M, et al. Adherence to medication among Parkinson’s disease patients using the adherence to refills and medications scale. Int J Clin Pract. 2022;2022:6741280. https://doi.org/10.1155/2022/6741280
- Mowlem FD, Elash CA, Dumais KM, Haenel E, O’Donohoe P, Olt J, et al. Best practices for the electronic implementation and migration of patient-reported outcome measures. Value Health. 2024;27(1):79–94. https://doi.org/10.1016/j.jval.2023.10.007
- Patruno C, Napolitano M, Ferrucci S, Brambilla L, Hansel K, Corazza M, et al. Four-week urticaria activity score-7 as a useful patient-reported outcome to assess chronic spontaneous urticaria: a multicentre study evaluation of adherence and patients’ perspective. Acta Derm Venerol. 2019;99(10):903–4. https://doi.org/10.2340/00015555-3231
- Daniëls NEM, Hochstenbach LMJ, Van Zelst C, Van Bokhoven MA, Delespaul PAEG, Beurskens AJHM. Factors that influence the use of electronic diaries in health care: scoping review. JMIR Mhealth Uhealth. 2021;9(6):e19536. https://doi.org/10.2196/19536
- Meyer VM, Benjamens S, Moumni ME, Lange JFM, Pol RA. Global overview of response rates in patient and health care professional surveys in surgery: a systematic review. Ann Surg. 2022;275(1):e75–81. https://doi.org/10.1097/SLA.0000000000004078
- Edwards P, Perkins C. Response is increased using postal rather than electronic questionnaires – new results from an updated Cochrane Systematic Review. BMC Med Res Methodol. 2024;24(1):209. https://doi.org/10.1186/s12874-024-02332-0
- Stone AA, Shiffman S, Schwartz JE, Broderick JE, Hufford MR. Patient compliance with paper and electronic diaries. Contr Clin Trials. 2003;24(2):182–99. https://doi.org/10.1016/S0197-2456(02)00320-3
- Zeng Q, Chen W, Xu S, Yang X, Xue D, Peng Y, et al. Understanding facilitators and barriers to adherence in home-based pulmonary rehabilitation for chronic obstructive pulmonary disease patients using the Health Belief Model: a qualitative study. Front Public Health. 2025;13:1553744. https://doi.org/10.3389/fpubh.2025.1553744
- Anisha SA, Sen A, Ahmad B, Bain C. Exploring acceptance of digital health technologies for managing non-communicable diseases among older adults: a systematic scoping review. J Med Syst. 2025;49(1):35. https://doi.org/10.1007/s10916-025-02166-3
- Collen MF, Miller RA. The early history of hospital information systems for inpatient care in the United States. In: Collen MF, Ball MJ, editors. The history of medical informatics in the United States [Internet]. London: Springer London; 2015, p. 339–83 [cited 2025 Sep 8]. (Health Informatics). Available from: https://link.springer.com/10.1007/978-1-4471-6732-7_6
- Engle RL. The evolution, uses, and present problems of the patient’s medical record as exemplified by the records of the New York Hospital from 1793 to the present. Trans Am Clin Climatol Assoc. 1991;102:182–9; discussion 189–192.
- Schatz M, Meltzer EO, Nathan R, Derebery MJ, Mintz M, Stanford RH, et al. Psychometric validation of the rhinitis control assessment test: a brief patient-completed instrument for evaluating rhinitis symptom control. Ann Allergy Asthma Immunol. 2010;104(2):118–24. https://doi.org/10.1016/j.anai.2009.11.063
- Młynek A, Zalewska-Janowska A, Martus P, Staubach P, Zuberbier T, Maurer M. How to assess disease activity in patients with chronic urticaria? Allergy. 2008;63(6):777–80. https://doi.org/10.1111/j.1398-9995.2008.01726.x
- Chan RJ, Crichton M, Crawford-Williams F, Agbejule OA, Yu K, Hart NH, et al. The efficacy, challenges, and facilitators of telemedicine in post-treatment cancer survivorship care: an overview of systematic reviews. Ann Oncol. 2021;32(12):1552–70. https://doi.org/10.1016/j.annonc.2021.09.001
- Mammen JR, Schoonmaker JD, Java J, Halterman J, Berliant MN, Crowley A, et al. Going mobile with primary care: smartphone-telemedicine for asthma management in young urban adults (TEAMS). J Asthma. 2022;59(1):132–44. https://doi.org/10.1080/02770903.2020.1830413
- Vieira RJ, Sousa-Pinto B, Cardoso-Fernandes A, Jácome C, Portela D, Amaral R, et al. Control of allergic rhinitis and asthma test: a systematic review of measurement properties and COSMIN analysis. Clin Transl Allergy. 2022;12(9):e12194. https://doi.org/10.1002/clt2.12194
- Simoni L, Recenti F, Roncari B, Tanzini L, Bagnardi V, Fiori G, et al. The authorization process of observational studies in Italy: exploring two decades of Ethics Committee approval data. Ann Ist Super Sanita. 2025;61(2):109–15.
- Guckian J, Thampy H. Gaining ethics approval for health professions education research. Clin Exp Dermatol. 2022; 47(12):2081–4. https://doi.org/10.1111/ced.15185
- Parkinson L. Why is ethical approval important? Aust J Ageing. 2022;41(3):355–6. https://doi.org/10.1111/ajag.13125
Copyright Ownership: This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0. The authors of this article own the copyright.
Appendix
Appendix B. Technical description of the digital medical record management system
Once the data have been entered into the interface, the patient’s medical record is generated in a digital system (made in Python).
In the medical record, a folder specific to the patient’s pathology will be created, containing a file with RCAT or UAS7.
The patient’s medical record is named using the patient’s first and last name. This record is created directly on a corporate cloud storage service managed by the “M. Albanesi” Allergy and Immunology Unit’ and contains the file related to the patient’s pathology.
For access, free URL shortening services are used to convert a URL into a shorter link, composed of the patient’s ID. This approach allows the patient to access their medical file through a personalized link.
The process begins with the patient entering their first and last name in the telemedicine section of the website. These data serve as input to generate the shortened link that points to the Excel file (to be filled out) related to their medical record. This link simplifies direct and secure access to the Excel document.
The latter is accessible to both the doctor and the patient at the same time, allowing them to make changes in real time. The patient enters their data (name and surname) in the dedicated section of the website. In fact, thanks to WordPress, the patient has the ability to open their personal Excel file, which is contained exclusively in their medical record folder.
The system’s cybersecurity is ensured by the advanced solutions provided by cloud technology.
Appendix C. Adherence trends in prolonged telemedicine therapy for chronic urticaria and chronic rhinitis
Prolonged Therapy – Second Compilation
For some patients, continuation of therapy was required. Therefore, within the same data acquisition timeframe, it was necessary, due to clinical needs, for some patients with previously diagnosed urticaria and chronic rhinitis to continue their therapy. In this context, telemedicine remained essential for ensuring the physician’s observation of the data. Consequently, second therapy diaries were provided to 24 patients (24% of the initial 100 patients).
Specifically, 22 out of these 24 patients (91.66% of the 24) were asked to complete the UAS for an additional 30 days.
Of these, 73% adhered to telemedicine, while 27% did not adhere. Conversely, the remaining 2 patients (8.34% of the 24) were asked to complete the RCAT, but none completed it. This trend is highlighted by the data presented in Supplemental Figure 1.
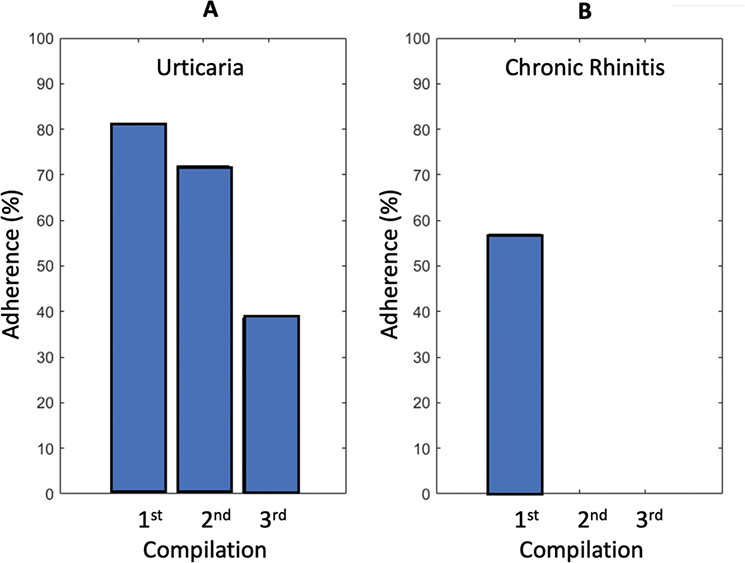
Supplemental Fig. 1. Percentage of telemedicine adherence in patients with chronic urticaria (A) and chronic rhinitis (B).
For both conditions, the Figure shows the percentage of patients who adhered to telemedicine in relation to the different cycles of questionnaire completion, divided into:
-First completion: represents the adherence rate to telemedicine for patients who completed the questionnaire after receiving the prescribed therapy following the first visit, that is, in the first month of treatment;
-Second completion: indicates the percentage of patients who adhered to telemedicine after receiving an additional therapeutic prescription following the second visit, that is, during the second month of treatment;
-Third completion: represents the percentage of patients who adhered to telemedicine after receiving a new prescribed therapy during the third visit, that is, in the third month of treatment.
Among all patients who also completed the second diary (regardless of whether they had chronic rhinitis or urticaria), due to clinical needs, some were required to further continue their therapy by completing a third questionnaire via telemedicine.
In particular, this was necessary (according to the physician) for 14 of the previously mentioned 24 patients (58.33% of the 24).
Of these:
-93% were asked to complete the UAS, being affected by urticaria. Among these, 38.5% chose to complete the diary, while 61.5% did not adhere;
-The remaining 7%, that is, one patient, was asked to complete the RCAT, being affected by chronic rhinitis. However, this patient did not complete it.
Prolonged Therapy – Third Compilation
For other patients, a third cycle of treatment was necessary. Only 38.5% (5 patients) with urticaria completed the diary, while 61.5% (9 patients) did not adhere to it.
Similarly, the patient with chronic rhinitis who was supposed to complete the RCAT did not adhere to completing the diary.
An evident decrease in the adherence rate to telemedicine was observed with an increasing number of treatment cycles. Specifically, the data show that adherence is highest during the first month of treatment, then decreases in the second month and further declines in the third month. This trend was evident in patients with both urticaria and chronic rhinitis, as illustrated in Supplemental Figure 1.
These results indicate a trend of decreasing adherence to telemedicine with the progression of treatment cycles, suggesting the need for targeted strategies to improve patient participation in prolonged treatments.
Appendix D. Associations Between Telemedicine Adherence and Demographic/Educational Characteristics
To assess the possible association between adherence to telemedicine and demographic/educational characteristics, we performed a series of statistical comparisons both within the individual cohorts (urticaria and rhinitis under immunotherapy) and between the two cohorts.
First, we conducted an analysis within the two groups (urticaria cohort and rhinitis-under-immunotherapy cohort) applying Student’s t-tests to age: in the urticaria group the comparison between adherent (mean age 35.27 ± 10.13 years) and non-adherent (mean age 28.53 ± 11.69 years) patients shows a statistically significant difference (P = 0.036082); whereas, in the immunotherapy group the comparison between adherent (mean age 29.88 ± 8.92 years) and non-adherent (mean age 31.25 ± 13.77 years) patients does not show statistically significant differences (P = 0.816063). A detailed summary of these comparisons is provided in Supplemental Table 1.
Subsequently, we evaluated the differences between groups (urticaria vs immunotherapy), considering adherent and non-adherent patients separately and employing Student’s t-tests for age: among adherent patients only, the comparison urticaria (mean age 35.27 ± 10.13 years) versus immunotherapy (mean age 29.88 ± 8.92 years) does not show statistically significant differences (P = 0.164707); similarly, among non-adherent patients only, urticaria (mean age 28.53 ± 11.69 years) versus immunotherapy (mean age 31.25 ± 13.77 years) does not show statistically significant differences (P = 0.622635). A detailed summary of these comparisons is provided in Supplemental Table 2.