ORIGINAL RESEARCH
Digital Transformation in Oncology: Monitoring of Adverse Effects of Oral Antineoplastic Agents Through mHealth Solutions
Carlota de Miguel Barbero, RN, PhD1,2, Elvira Gea Rodríguez PharmD, PhD1,3 and Francesc Garcia Cuyàs, MD, PhD1,4
1Department of Research, University of Andorra, Health Sciences and Healthcare Services Research Group, Andorra; 2International Doctoral School, Doctor in Nursing, Member of the Health Sciences and Healthcare Services Research Group, University of Andorra, Andorra; 3International Doctoral School, Doctor in Pharmacy, Coordinator of the Health Sciences and Healthcare Services Research Group, University of Andorra, Andorra; 4International Doctoral School, Doctor in Medicine, Adjunct Professor, and Member of the Health Sciences and Healthcare Services Research Group, University of Andorra, Andorra.
Keywords: cancer, digital transformation, health empowerment, medical oncology, mHealth, mobile apps, nursing, pharmacy, telehealth, toxicity
Abstract
Background: Healthcare systems face sustainability challenges due to the growing burden of chronic diseases such as cancer. The increasing use of oral antineoplastic agents shifts part of treatment management to the home setting, where adverse effects might go undetected between clinical visits.
Objective: The authors aimed to redesign the care process for monitoring adverse effects related to oral antineoplastic agents through a mobile health application and to evaluate its implementation in routine outpatient oncology care.
Methods: A prospective interventional study was conducted in adult oncology patients receiving oral antineoplastic agents. All participants received standardized training on symptom identification and application use. Participants used a mobile health application to report adverse effects from home over 3 months. Quantitative data on adverse effects, application use, and patient-reported outcomes were collected, together with qualitative feedback on user experience.
Results: Forty-seven patients were included. The most frequently reported adverse effects were asthenia, abdominal pain, and anxiety. A small proportion of severe adverse effects required referral to other healthcare professionals. Most patients (87.2%) were classified as intermediate technology users. Application quality was rated highly, with a mean score of 4.44 out of 5. An increase in empowerment classification was observed, mainly related to improved preparation for clinical consultations.
Conclusions: Integrating a mobile health application into outpatient oncology care is feasible and supports timely monitoring of adverse effects and patient–professional communication. Improvements in empowerment-related behaviors were modest and primarily associated with patient engagement.
Plain Language Summary
Oral antineoplastic agents allow many patients with cancer to continue treatment at home, but adverse effects may appear between hospital visits. This prospective interventional study evaluated whether a mobile health app could support symptom monitoring and communication with healthcare professionals. Forty-seven patients in Andorra used the Abeona Health app for 3 months to report symptoms and contact the care team. Most patients reported adverse effects, and app use was associated with improved patient empowerment and communication. These findings suggest that mHealth tools may support safer and more patient-centered oncology follow-up.
Citation: Telehealth and Medicine Today © 2026, 11: 627
DOI: https://doi.org/10.30953/thmt.v11.627
Copyright: © 2026 The Authors. This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See http://creativecommons.org/licenses/by-nc/4.0. The authors of this article own the copyright.
Submitted: September 8, 2025; Accepted: February 19, 2026, Published: March 28, 2026
Corresponding Author: Carlota de Miguel Barbero, Email: cmiguelb@uda.ad
Competing interests and funding: All authors declare no conflicts of interest to disclose.
This article was funded by the University of Andorra.
Digital transformation refers to the strategic redesign of healthcare processes through the use of interoperable digital tools, data-driven workflows, and patient-centered communication.1 In the context of healthcare systems facing increasing pressure from chronic diseases, digital transformation is identified as a key enabler to improve efficiency, sustainability, and quality of care.2–4 European and international health strategies emphasize the role of digital health technologies to empower patients, optimize resource use, and support more proactive and personalized models of care.5
Cancer is increasingly managed as a chronic condition, requiring long-term treatment and close monitoring.6 The growing use of oral antineoplastic agents has shifted part of treatment administration and monitoring from hospital settings to patients’ homes.7 While this approach offers greater convenience and autonomy, it also introduces safety challenges, particularly related to the timely identification and management of adverse effects.8 In routine practice, adverse effects are often reported during scheduled clinical visits, which may delay intervention and negatively affect patient outcomes.
Mobile health applications offer a potential solution by enabling real-time reporting of symptoms and direct communication between patients and healthcare professionals. When integrated into care processes, these tools may support earlier detection of adverse effects, improve coordination of care, and enhance patient engagement. However, evidence on the real-world implementation of such tools in oncology, particularly within routine pharmaceutical care workflows, is limited.9–13
The objective of this study is to redesign the pharmaceutical care process for monitoring adverse effects of oral antineoplastic agents through the implementation of a mobile health application in a real-world clinical setting. The study focused on the feasibility of integrating the application into routine practice and on evaluating patient engagement, user experience, application quality, and empowerment-related outcomes.
Material and Methods
Study Design, Setting, and Population
A prospective interventional study was conducted to redesign and evaluate the pharmaceutical care process for monitoring adverse effects associated with oral antineoplastic agents through a mobile health application, using a pragmatic real-world implementation approach.
The study was carried out in the Principality of Andorra within the outpatient pharmacy service of the Andorran Health Care Service. Adult oncology patients (≥ 18 years) receiving oral antineoplastic agents dispensed by the SAAS were eligible for inclusion.
Inclusion and Exclusion Criteria, Recruitment, and Follow-up
The criteria for participation included adult patients receiving oral antineoplastic agents, their ability to use a smartphone after training, and provision of written informed consent.
Exclusion criteria included language barriers, cognitive impairment, visual impairment, Eastern Cooperative Oncology Group performance status ≥ 3,14 Karnofsky performance score ≤ 50,15 and incompatible availability during the study (e.g. treatment completion or leaving the country).
Patients were recruited consecutively at the outpatient pharmacy clinic between November 2022 and May 2023. Eligible patients received oral and written information about the study from the principal investigator and the oncology pharmacist. After providing informed consent, participants were enrolled chronologically.
Each participant was followed for up to three months, aligned with their pharmacological regimen. The overall study duration was 9 months, including follow-up of participants recruited at the end of the inclusion period.
Study Phases
The selection of the mobile app included a systematic search for mobile applications across major commercial application stores, including Google Play, App Store, and Amazon Appstore. Applications were evaluated using quality criteria derived from the iSYScore methodology16 and the accreditation framework of the TIC Salut Social Foundation.17
Reengineering the pharmaceutical care process for outpatient treatment with the Office of Aging and Adult Services (OAAS) at the SAAS Incorporating Digital Tools focused on the pharmaceutical care process being redesigned using a co-creation approach involving all SAAS pharmacists through a structured working session and strengths, weaknesses, opportunities, and threats (SWOT) analysis.18
Regarding the implementation of the mobile app, the study implementation was structured into the following four key sections:
First, sociodemographic and clinical data were collected, including sex, age, tumor type, treatment status (initiation or continuation), and use of concomitant parenteral antineoplastic agents. Reported adverse effects were reviewed daily by the principal investigator in collaboration with the oncology pharmacist and lead oncology nurse, who intervened when medical attention was required. Severity grading was performed according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0.19
Second, patients and caregivers were assessed for their ability to use smartphones and willingness to use an app for active disease management (empowerment) if recommended by a healthcare professional.
Participants’ digital competence was classified using a pragmatic three-level operational rubric (basic, intermediate, advanced) developed by the research team to tailor training and support according to participants’ smartphone use skills.
Third, all participants received standardized training focused on digital literacy and health literacy. Training was adapted to participants’ smartphone use skills and included application installation, navigation, and symptom identification.
Fourth, during follow-up, participants used the mobile health application from home to report adverse effects related to oral antineoplastic agents. Adverse effects were recorded using structured electronic forms available in the application, with predefined symptom categories and severity levels. Participants could also report symptoms not included in predefined lists through free-text entries using the messaging function.20
Reported adverse effects were transmitted in real time to the healthcare team. Messages and symptom reports were reviewed daily by the principal investigator in collaboration with the oncology pharmacist and lead oncology nurse. When clinically indicated, remote advice, additional follow-up, or referral to other healthcare professionals was initiated according to established clinical criteria.
Analysis of User Experience with the App, Including Quality from the Patient Perspective, and Empowerment in Health Management
Following a literature review, an analysis of commonly used mHealth questionnaires was conducted to identify the most suitable tools for this research context. Selection criteria included scientific validity, short response time to minimize patient burden, Spanish-language availability, and alignment with study objectives such as adverse effect management and patient–healthcare professional communication.
Usability and perceived app quality from the patient perspective were assessed using the user version of the Mobile Application Rating Scale (uMARS),21 which integrates objective domains (engagement, functionality, aesthetics, and information quality) with subjective evaluation of perceived impact. The uMARS was selected due to its validated Spanish version and suitability for evaluating mHealth applications in real-world clinical settings.
Empowerment in health management was assessed using the classification proposed by the National Observatory of Technology and Society (ONTSI),22 chosen for its simplicity, brief administration time, and alignment with the Spanish healthcare context. This classification focuses on patient engagement behaviors relevant to digital health use.
All questionnaires were self-administered by participants at the end of the 3-month follow-up. Descriptive statistics were used to summarize scores.
Ethical Considerations and Study Funding
The study was approved by the Clinical Research Ethics Committee of the Andorran Health Care Service (SAAS) (approval number: 11-2021-CEIC-ONCOTIC; approval date: 10 June 2021) ensuring compliance with the ethical principles of biomedical research. Written informed consent was obtained from all participants prior to inclusion. Data confidentiality and participant anonymity were ensured throughout the study in compliance with applicable data protection regulations.
Statistical Analysis
Statistical analyses were performed using Jamovi. Descriptive statistics were used to summarize the data. Comparisons were conducted using analysis of variance (ANOVA), with statistical significance set at I < 0.05.
Descriptive statistics were used to summarize sociodemographic characteristics, app use, adverse effect reporting, and patient-reported outcomes. Continuous variables are presented as means and standard deviations, and categorical variables as frequencies and percentages.
Results
Selection of the Mobile App
After evaluating applications available on Google Play, the App Store, and Amazon Appstore, 12 applications met the study inclusion criteria: Abeona Health®, Cancer.net®, LivingWith Cancer Support®, Oncomed®, e-Onco Salud®, ICOnnecta®, MedWatcher®, ChemoWave for Cancer Patients®, MedClin®, iCancer Health®, Noona®, and Cancergraph®. Applications intended exclusively for healthcare professionals or lacking interactive functionality were excluded.
Using two structured evaluation frameworks, eight applications were prioritized: e-Onco Salud®, ICOnnecta®, Cancer.net®, ChemoWave for Cancer Patients®, LivingWith Cancer Support®, MedWatcher®, Noona®, and iCancer Health®. Evaluation scores ranged between 31 and 32 out of 40 for one framework and between 31 and 36 out of 47 for the other. e-Onco Salud® achieved the highest scores; however, it was excluded due to maintenance costs and lack of available funding.
Consequently, Abeona Health® (Athenea Solutions & Tech SL) was selected. Despite lower evaluation scores, the application offered free-of-charge maintenance and monitoring of seven common adverse effects: hypertension, vomiting, diarrhea, hand–foot syndrome, mucositis, nervous system disorders, and fever. The application was available in Spanish and enabled direct communication with healthcare professionals via messaging and telephone.
Following publication of a previous study evaluating the application, six additional adverse effects were incorporated: anxiety, asthenia, abdominal pain, constipation, nausea, and skin toxicity. Clinical decision-support algorithms and data protection adaptations resulted in an application version tailored to the Andorran healthcare context.
Reengineering the Pharmaceutical Care Process for Outpatient Treatment with OAAS at the SAAS Incorporating Digital Tool
The SWOT analysis identified several strengths supporting implementation of a digital adverse-effect monitoring tool. These include availability of a dedicated mobile application an established oral antineoplastic agent dispensing protocol adaptable to process reengineering, institutional authorization, real-time pharmaceutical team support, and high national smartphone integration.
Identified weaknesses included small sample size, reliance on active patient participation, lack of integration with the electronic health record, and potential digital barriers among older patients. Threats included potential patient dropout, limited smartphone availability in some patients, and failure to achieve expected outcomes. Opportunities highlighted the transition toward a proactive digital care model, enabling real-time adverse-effect reporting, direct patient–healthcare professional communication, and standardized safety processes.
Based on these findings, consensus was reached to integrate the digital tool at the patient education stage of the pharmaceutical care process. Reengineering this step resulted in a redesigned care pathway that incorporated real-time adverse-effect monitoring and bidirectional communication between patients and healthcare professionals. The redesigned pharmaceutical care pathway is illustrated in Figure 1.
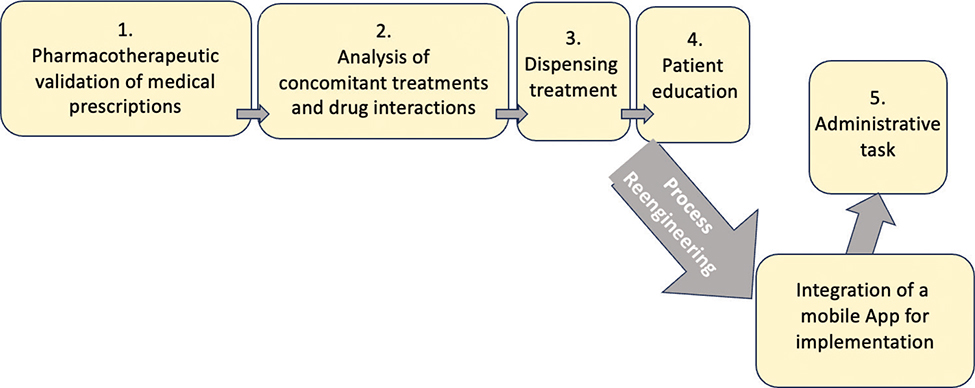
Fig. 1. Reengineering pharmacy care management process for the Office of Aging and Adult Services (OAAS) treatment incorporating digital tools. OAAS: Office of Aging and Adult Services.
Source: Adapted with permission from Carlota de Miguel, 2024, from “Digital transformation of the oncology care model for monitoring the adverse effects of oral antineoplastic treatment. 2024.”
Implementation of the Mobile App
Data were collected from 93 individuals who were screened during recruitment. Of these, 30 did not meet the inclusion criteria, and 16 declined to participate. Reasons for their refusal included lack of time, limited motivation for daily data recording, and difficulties related to digital literacy.
The final study sample consisted of 47 participants. Among female participants (n = 27; 57.4%), the most frequent oncologic diagnoses were breast cancer (21.3%), rectal cancer (6.4%), and chronic lymphocytic leukemia (6.4%). Among male participants (n = 20; 42.6%), the most common diagnoses were colon cancer (8.6%), chronic myeloid leukemia (6.4%), and chronic lymphocytic leukemia (6.4%). The distribution of participants by cancer type and sex is shown in Table 1 (percentages are calculated using the total study sample as the denominator (n = 47). Other cancer diagnoses accounted for the remaining participants.
No statistically significant differences in application use or engagement were observed across cancer types or between sexes (ANOVA, p > 0.05).
The most frequently prescribed oral antineoplastic agents were capecitabine, palbociclib, ibrutinib, and imatinib. Seven patients (14.9%) received combination therapy with parenteral antineoplastic agents. Within the pharmaceutical care service, initial consultations accounted for 25.5% (n = 12) of visits, while follow-up visits represented 74.5% (n = 35).
The Ability of Patients and Caregivers to Use Smartphones and Willingness to Adopt an App for Active Disease Management (Empowerment) if Recommended by a Healthcare Professional
Most participants owned a personal smartphone (89.3%), while 10.7% used a device belonging to a family member. Although smartphone access was widespread, only 34% reported using health-related applications prior to the study.
Regarding communication preferences, 66% preferred telephone contact with healthcare professionals, while 25.5% favored mobile applications. All participants expressed willingness to use a mobile application if recommended by a healthcare professional, and none expressed willingness to pay for application access.
Based on the operational digital competence classification, 87.2% of participants were classified as intermediate users and 12.8% as basic users. No advanced users were identified.
Training Patients to Use the App
All participants received standardized training focused on digital and health literacy. Training was adapted to digital competence level. During training, 23 queries from nine participants were addressed, including clinical questions related to symptom identification (56.5%) and technical questions related to application functionality (43.5%). All issues were resolved during training sessions or follow-up support.
Use of App by Outpatients to Monitor and Report Adverse Effects
During the 9-month study period, 41 participants (87.2%) reported at least one adverse effect, while six (12.8%) reported no symptoms. A total of 213 adverse effects were recorded, with a mean of 5.53 events per patient (standard deviation ± 3.60; range 1–14).
Of all reported events, 77.0% (n = 164) corresponded to predefined symptoms included in the application, while 23.0% (n = 49) were reported through free-text messaging, including symptoms such as myalgia, muscle cramps, and weight loss. Percentages represent the proportion of each adverse-effect event relative to the total number of reported adverse effects (n = 213) (Figure 2). Individual patients could report more than one adverse effect; therefore, percentages do not reflect patient-level prevalence and do not sum up to 100%.
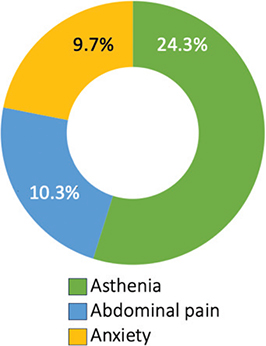
Fig. 2. Most frequently reported adverse effects from the app’s predefined list.
Source: Adapted with permission from Carlota de Miguel, 2024, from “Digital transformation of the oncology care model for monitoring the adverse effects of oral antineoplastic treatment. 2024.”
A small portion of CTCAE v5.0 grade 3 adverse events required referral to other healthcare professionals following clinical assessment, and one adverse event resulted in hospital admission.
The use of the messaging function was observed in all participants. A total of 966 messages were exchanged, with a mean of 20.4 messages per patient (standard deviation ±13.6; range 2–87). Messages related to adverse effect reporting accounted for 48.9% of communications, while 47.6% confirmed absence of symptoms. Requests for oncology appointments represented 2.7% of messages, and pharmacotherapeutic or technical queries accounted for less than 1%.
At baseline, 74.4% of participants were classified as empowered, with a mean empowerment score of 3.31. After 3 months of application use, the proportion classified as empowered increased to 87.2%, while the mean score remained unchanged.
Repeated measures analysis showed a statistically significant improvement in preparation for medical consultations, with a weak effect size. No significant differences in empowerment outcomes were observed by sex or age.
Application quality was evaluated by 46 of 47 participants (97.8%). The mean overall quality score was 4.44 out of 5 (95% confidence interval: 4.35–4.52), while subjective quality scored 4.42 out of 5 (95% confidence interval: 4.28–4.56). No significant differences in quality ratings were observed by sex or age.
Qualitative feedback highlighted themes related to personalized care, satisfaction with application use, direct communication with healthcare professionals, and support for self-management of adverse effects.
Discussion
This study describes the real-world implementation of a mobile health app to support monitoring of adverse events associated with oral antineoplastic agents in routine outpatient oncology care. The findings demonstrate that integrating a digital tool into the pharmaceutical care process is feasible and supports structured symptom reporting, timely follow-up, and sustained communication between patients and healthcare professionals, in line with current digital health strategies in oncology and chronic disease management.1–4,23–25
The selection of the app was guided by established digital health evaluation frameworks to ensure usability, safety, and suitability for clinical use.16,17,26 Although multiple oncology-related apps are available, previous studies highlighted that many lack clinical validation, interoperability, or bidirectional communication functionalities.27–29 In this context, the final selection prioritized pragmatic feasibility, institutional integration, and sustainability over maximal technical scoring, reflecting real-world constraints commonly encountered in healthcare system implementation initiatives.30–32
Reengineering the pharmaceutical care process to incorporate digital monitoring addressed a relevant safety gap in conventional follow-up of patients treated with oral antineoplastic agents, where adverse effects are frequently underreported or reported retrospectively during scheduled visits.9,33 By enabling real-time reporting from the home setting, the app facilitated early identification and remote management of most adverse effects, with a limited proportion of referrals and hospital admissions. These findings are consistent with previous evidence suggesting that digital symptom monitoring can support proactive care models, improve continuity of care, and enhance patient safety in oncology settings.9,20,24,25,33,34
Patient engagement with the app was high, as reflected by universal use of the messaging function and sustained interaction throughout the study period. Similar levels of engagement have been reported in other mHealth interventions when digital tools are integrated into routine care pathways and supported by healthcare professionals.4,6,26,32 Importantly, engagement in this study was observed even among participants with limited prior experience using health-related apps, suggesting that targeted digital and health literacy training embedded within pharmaceutical care may mitigate commonly reported barriers related to age or digital skills.5,27,35
With respect to empowerment-related outcomes, improvements were primarily observed in specific aspects and were mainly driven by enhanced preparation for medical consultations. While the proportion of participants classified as empowered increased over time, the overall empowerment score remained stable, and changes were concentrated in a single behavioral domain. This finding underscores the importance of cautious interpretation and aligns with previous studies indicating that short-term digital interventions are more likely to influence specific engagement behaviors rather than produce broad changes in patient empowerment.36–38 Avoiding overinterpretation of empowerment outcomes is particularly relevant in implementation-focused studies, where feasibility and process optimization are primary objectives.39
App quality and user experience were rated highly using the uMARS instrument, with scores comparable to those reported in evaluations of mHealth tools in oncology and other chronic conditions.40–42 Qualitative feedback further highlighted the perceived value of personalized care, reassurance provided by continuous professional support, and direct communication with healthcare professionals. These dimensions have been consistently identified as key determinants of acceptability, trust, and sustained use of digital health interventions.
Scalability of the Model and Future Lines
The care model described in this study was implemented within a small national healthcare system; however, its underlying structure is potentially scalable to other settings. The use of a mobile health app integrated into the pharmaceutical care process relies on elements that are transferable to larger healthcare systems, such as standardized adverse event reporting, remote follow-up, and bidirectional communication between patients and healthcare professionals.
Scalability would require adaptation to local organizational structures, integration with electronic health records, and alignment with regulatory and data protection frameworks. Future lines of research should explore long-term clinical outcomes, integration with other digital health platforms, and evaluation of cost-effectiveness. Additionally, assessing the impact of this model in larger and more heterogeneous populations would provide further evidence to support broader implementation.
Limitations
This study has several limitations. First, the sample size was relatively small; however, it reflects the entire national population of patients receiving oral antineoplastic agents within the Andorran Health Care Service during the study period. Second, the study was conducted within a single healthcare system, which may limit the generalizability of the findings to other settings with different organizational structures or levels of digital maturity. Third, the follow-up period was limited to three months per participant, precluding evaluation of long-term outcomes such as adherence, quality of life, or healthcare utilization. Finally, the absence of a control group limits causal inference, as the study was designed to evaluate feasibility and real-world implementation rather than comparative effectiveness.
In addition, the app selection process was restricted to applications available on the main commercial platforms (Google Play, App Store, and Amazon Appstore), and the evaluation relied on two established assessment frameworks. Although these frameworks are widely used and appropriate for the study objectives, other evaluation models or platforms not included in the analysis may have identified different tools. Finally, the absence of a control group limits causal inference, as the study was designed to assess feasibility and real-world implementation rather than comparative effectiveness.
Despite these limitations, this study provides relevant real-world evidence on the integration of a mobile health app into pharmaceutical care for oncology patients treated with oral antineoplastic agents. The findings support the role of digital tools as complementary components of patient-centered care models, particularly for enhancing communication, supporting safe outpatient management of adverse effects, and strengthening continuity of care. Future research should focus on long-term outcomes, scalability, and integration with electronic health records to further consolidate the evidence base for digital transformation in oncology care.2,3,5,11,40
Conclusion
The implementation of a mobile health app for monitoring adverse effects associated with oral antineoplastic agents was feasible and successfully integrated into routine pharmaceutical care. The model supported timely reporting of adverse effects, facilitated continuous communication between patients and healthcare professionals, and contributed to continuity of care in the outpatient oncology setting. Improvements in patient engagement were observed, although changes in empowerment-related outcomes were modest and mainly related to preparation for clinical consultations. These findings support the role of digital tools as complementary components of patient-centered oncology care and provide a basis for future studies evaluating scalability and long-term impact.
Contributions
Carlota de Miguel Barbero led the study, collected and analyzed the data, and drafted the manuscript. Elvira Gea Rodríguez and Francesc Garcia Cuyàs contributed to study design, interpretation of findings, and critical revision of the manuscript. All authors approved the final manuscript.
Data Availability Statement (DAS), Data Sharing, Reproducibility, and Data Repositories
The data generated and analyzed during this study were anonymized and may be made available from the corresponding author upon reasonable request.
Application of AI-Generated Text or Related Technology
Artificial intelligence tools were used to assist with language editing and clarity of expression. These tools did not generate original scientific content, data, analyses, or conclusions. All content was reviewed, validated, and approved by the authors, who take full responsibility for the manuscript.
Acknowledgments
The authors acknowledge the University of Andorra for providing the academic framework and research opportunity, with special thanks to Dr. Aleix Dorca-Josa for statistical support. The authors also thank the Andorran Health Care Service (SAAS) for facilitating the pilot implementation within the national healthcare system. Additional acknowledgement is extended to the Official Nursing College of Andorra for partial doctoral funding and to Athenea Solutions for providing the study application under free licensing conditions. The authors are especially grateful to the patients who altruistically participated in the study and contributed their time and experience to support the improvement of oncology care.
References
- SEIS, AMETIC, FENIN. Hacia la transformación digital del sector salud: 10 medidas para su impulso [Internet]. Spain: SEIS–AMETIC–FENIN; 2016 [cited Sep 6]. Available from: https://lc.cx/U7V3gO
- European Commission. eHealth action plan 2012–2020: innovative healthcare for the 21st century [Internet]. Brussels; 2012 [cited Sep 6]. Available from: https://bit.ly/2KgpFj6
- Hammer M. Reengineering work: don’t automate, obliterate. Harv Bus Rev. 1990;68(4):104–12.
- Hammer M, Champy J. Reengineering the company: a manifesto for business revolution. Bus Horiz. 1993;36(5):90–1.
- World Health Organization. Global strategy on digital health 2020–2025 [Internet]. Geneva: WHO; 2021 [cited Sep 6]. Available from: https://www.who.int
- Garcia-Cuyàs F, de San Pedro M, Roldán Martínez J. La salud digital como motor de cambio hacia nuevos modelos asistenciales. Med Clin (Barc). 2015;145:38–42.
- Siefert ML, Blonquist TM, Berry DL, Hong F. Symptom-related emergency department visits during ambulatory cancer treatment. J Community Support Oncol. 2015;13(5):188–94.
- Mirkovic J, Kaufman DR, Ruland CM. Supporting cancer patients in illness management: usability evaluation of a mobile app. JMIR Mhealth Uhealth. 2014;2(3):e33.
- Klasnja P, Hartzler A, Powell C, Pratt W. Supporting cancer patients’ information management with mobile technology. AMIA Annu Symp Proc. 2011;2011:732–41.
- De Miguel Barbero C, Avellanet Viladomat M, Gea Rodríguez E, Garcia-Cuyàs F. Análisis descriptivo de aplicaciones móviles para el seguimiento de efectos adversos de antineoplásicos. Rev Esp Salud Publica. 2022;96:e202201003.
- De Miguel Barbero C, Gea Rodríguez E, Garcia-Cuyàs F. Utilidad de la app Abeona Health® para la identificación de toxicidad inducida por quimioterapia. Rev Esp Salud Publica. 2023;97:e202312108.
- McCann L, Maguire M, Kearney N. Patients’ experiences using a mobile phone-based symptom management system. Eur J Cancer Care. 2009;18(4):394–424.
- Weaver A, Young AM, Rowntree J, Townsend N, Pearson S, Smith J, et al. Application of mobile phone technology for managing chemotherapy side effects. Ann Oncol. 2007;18(11):1888–92.
- Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5(6):649–55.
- ECOG-ACRIN Cancer Research Group. ECOG performance status scale [Internet]. 2022 [cited Sep 6]. Available from: https://ecog-acrin.org
- Grau I, Kostov B, Gallego JA, Grajales Iii F, Fernández-Luque L, Sisó-Almirall . Método de valoración de aplicaciones móviles de salud: índice iSYScore. Semergen. 2016;42(8):575–83.
- Fundació TIC Salut Social. Guía del proceso de acreditación de apps de salud [Internet]. Barcelona; 2017 [cited Sep 6]. Available from: https://ticsalutsocial.cat
- Promove Consultoria e Información SLNE. Xunta de Galicia. Cómo elaborar el análisis DAFO [Internet]. 2012 [cited Sep 6]. Available from: https://bit.ly/3OYMDMY
- U.S. Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) [Internet]. 2021 [cited Sep 6]. Available from: https://ctep.cancer.gov
- Govern d’Andorra. Estadística sobre equipamiento y uso de tecnologías de la información y comunicación en los hogares [Internet]. Andorra; 2024 [cited Sep 6]. Available from: https://lc.cx/KM5Y-H
- Martín-Payo R, arrasco-Santos S, Cuesta M, Stoyanov S, Gonzalez-Mendez X, Fernández Álvarez M. Spanish adaptation and validation of the user version of the Mobile Application Rating Scale (uMARS). J Am Med Inform Assoc. 2021;28(12):2681–6.
- Observatorio Nacional de las Telecomunicaciones y de la Sociedad de la Información. Los ciudadanos ante la e-Sanidad [Internet]. Spain; 2016 [cited Sep 6]. Available from: https://bit.ly/3wHnYGI
- Handa S, Okuyama H, Yamamoto H, et al. Effectiveness of a smartphone application as a support tool during chemotherapy. Clin Breast Cancer. 2020;20(3):201–8.
- Kearney N, McCann L, Norrie J, Taylor L, Gray P, McGee-Lennon M, et al. Evaluation of a mobile phone-based symptom management system. Support Care Cancer. 2009;17(4):437–44.
- Lettieri E, Fumagalli LP, Radaelli G, Bertele’ P, Vogt J, Hammerschmidt R, et al. Empowering patients through eHealth: a case report. BMC Health Serv Res. 2015;15:309.
- Norgaard O, Furstrand D, Klokker L, Karnoe A, Batterham R, Kayser L, et al. The eHealth literacy framework. Knowl Manag E-Learn. 2015;7(4):522–40.
- Vidal-Alaball J, Alarcon Belmonte I, Panadés Zafra R, Escalé-Besa A, Acezat Oliva J, Saperas Perez C. Digital transformation to reduce the digital divide. Aten Primaria. 2023;55(9):102731.
- Warrington L, Absolom K, Conner M, Kellar I, Clayton B, Ayres M, Velikova G. Electronic system for patients to report and manage side effects of cancer treatment: systematic review. J Med Internet Res. 2019;21(1):e10875.
- Berry DL, Blonquist TM, Nayak MM, Grenon N, Momani TG, McCleary NJ. Self-care support for cancer patients using iCancerHealth. Appl Clin Inform. 2018;9(4):833–40.
- Woods LS, Duff J, Roehrer E, Walker K, Cummings E. Patient experiences using a consumer mHealth app. JMIR Hum Factors. 2023;10:e13009.
- Mesko B. What can digital health bring to small countries? The Medical Futurist [Internet]. 2017 [cited Sep 6]. Available from: https://medicalfuturist.com
- Klasnja P, Hartzler A, Powell C, Phan G, Pratt W. Health weaver mobile: designing a mobile tool for managing personal health information during cancer care. AMIA Annu Symp Proc. 2010;2010:392–6.
- Rosa-Chavez G, Romero-Visurraga CA, Ramirez-Guardia E, Málaga G. Health literacy and treatment adherence in patients with hypertension. Rev Peru Med Exp Salud Publica. 2019;36(2):214–21.
- Collado Borrell R. Diseño e implementación de una aplicación móvil para el seguimiento farmacoterapéutico de pacientes en tratamiento con antineoplásicos orales [Doctoral thesis]. Madrid: Universidad Complutense de Madrid; 2019.
- SEOM. Cifras del cáncer en España 2025 [Internet]. Spain; 2025 [cited Sep 6]. Available from: https://lc.cx/uHQoTj
- Agustín Fernández MJ, Cajaraville Ordoña G, Carrera Soler MJ, et al. Nuevas tecnologías aplicadas al servicio de farmacia: proceso de uso de medicamentos citostáticos [Internet]. Spain: Astellas; 2014. Available from: https://n9.cl/wpkwa
- Arias López MDP, Ong BA, Borrat Frigola X, Fernández AL, Hicklent RS, Obeles , et al. Digital literacy as a new determinant of health: A scoping review. PLOS Digit Health. 2023;2(10):e0000279. https://doi.org/10.1371/journal.pdig.0000279
- Sotillos-González B, Buiza-Camacho B, Herrera-Usagre M, Escobar-Ubreva Á, Fernández-Bermejo MC, Santana-López V, et al. Citizen perspectives on mobile health prescription and ICT use in healthcare. J Healthc Qual Res. 2018;33(4):225–33.
- Ana B, Cristina C. SEOM. Toxicidad de los tratamientos oncológicos [Internet]. 2019 [cited Sep 6]. Available from: https://lc.cx/GwAyZk
- Andersen T, Ruland CM. Cancer patients’ questions and concerns expressed in an online nurse-delivered mail service. Stud Health Technol Inform. 2009;146:149–53.
- Ricart Martí P. Reingeniería de procesos para la mejora de la seguridad clínica en el servicio de medicina intensiva [Doctoral thesis]. Barcelona: Universitat Autònoma de Barcelona; 2016.
- Hospital Sant Joan de Déu. H2.0 Hospital Líquid [Internet]. Barcelona; 2019 [cited Sep 6]. Available from: https://lc.cx/fYbNu3
Copyright Ownership: This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See http://creativecommons.org/licenses/by-nc/4.0. The authors of this article own the copyright.