NARRATIVE/SYSTEMATIC REVIEWS/META-ANALYSIS
Empowering Parkinson’s Disease Management Through Mobile Applications: A Systematic Review and Meta-analysis
Mei Wah Lee, MSc  ; Umar Idris Ibrahim, PhD
; Umar Idris Ibrahim, PhD  ; and Pei Lin Lua, PhD
; and Pei Lin Lua, PhD 
Faculty of Pharmacy, Universiti Sultan Zainal Abidin (UniSzA), Terengganu, Malaysia
Keywords: mental health, mobile applications, Parkinson’s disease, physical function
Abstract
Introduction: Numerous mobile applications have been developed for Parkinson’s disease; however, their effectiveness in improving clinical outcomes is uncertain.
Objective: This systematic review evaluated the effectiveness of mobile applications for Parkinson’s disease in diagnosis and self-management.
Methods: A review of the literature was conducted using PubMed, Ovid Medline, Scopus, ScienceDirect, Cochrane Library, and EMBASE for articles related to mobile applications in Parkinson’s disease published between 2011 and November 10, 2024. Meta-analyses were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The quality of the selected studies was assessed.
Results: Nineteen studies involving 1,080 participants across 10 countries were included in the review. These studies included 15 cross-sectional studies and four randomized controlled trials. Mobile applications were used for symptom monitoring, medication reminders, exercise, rehabilitation, and diagnosis of Parkinson’s disease. A meta-analysis of four randomized controlled trials found no significant difference between mobile application interventions and control groups (overall standardized mean difference [SSMD] = –1.12, 95% confidence interval [CI]: –5.53 to 3.29, p = 0.62). Subgroup analyses revealed no significant effect for either motor (standard mean difference = 0.02, 95% CI: −0.41 to 0.46, p = 0.91) or nonmotor symptoms (standard mean difference = −777.73, 95% CI: −2306.27 to 750.80, p = 0.32).
Conclusion: Due to the limited number and heterogeneity of existing studies, the evidence regarding the effectiveness of mobile applications in Parkinson’s disease remains inconclusive. Further research is needed to strengthen the evidence and assess the clinical benefits of mobile applications in the management of Parkinson’s disease.
Citation: Telehealth and Medicine Today © 2026, 11: 676 - https://doi.org/10.30953/thmt.v11.676
DOI: https://doi.org/10.30953/thmt.v11.676
Copyright: © 2026 M.W. Lee et al. This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See http://creativecommons.org/licenses/by-nc/4.0. The authors of this article own the copyright.
Submitted: December 19, 2025; Accepted: January 1, 2026; Published: March 12, 2026
Corresponding Author: Umar Idris Ibrahim, Email: umaribrahim@unisza.edu.my
Competing interests and funding: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Parkinson’s disease (PD) is the second most common neurodegenerative disorder after Alzheimer’s disease. It involves the selective degeneration of dopaminergic neurons located in the substantia nigra of the midbrain.1,2 The number of patients with PD is estimated to increase to more than 9 million worldwide by 2030.3
Symptoms associated with PD vary among individuals, making PD extremely complex to treat. Motor symptoms include tremors, rigidity, postural instability, slowness of movement (bradykinesia), and uncontrollable movements (dyskinesia). Nonmotor symptoms include neuropsychiatric abnormalities, sleep disorders, autonomic dysfunction, and constipation.1,4
Patients with PD report a lower quality of life compared with healthy controls in most domains, particularly in physical function and mental health.5 Several factors, such as age, lack of knowledge, marital status, frequent hospitalization, duration of disease, severity of PD, anxiety and depression, ‘on-off’ phenomenon, and ‘wearing-off’ phenomenon, have a negative influence on quality of life in these patients.6–8 Previous research reveals that the rate of premature unemployment due to PD ranges from 27% to 70%. Older age, later disease onset, longer disease duration, greater severity of symptoms, and lack of support from coworkers are the main factors associated with giving up work.9
Patients with PD are admitted to hospitals at rates varying from 7% to 28% annually. Those suffering from PD or parkinsonism experience hospitalization approximately 1.5 times more often and typically remain hospitalized for a duration of 2 to 14 days longer than patients without PD.10 Patients with PD have higher rates of emergency hospital admissions with longer hospital stays, higher costs, and in-hospital mortality. A cross-sectional analysis of the English Hospital Episodes Statistics database from 2009 to 2013 revealed that during that time, there were 324,055 PD admissions among 182,859 patients, of which 232,905 were elective (72%).
The total cost was £907 million ($1,225 million) (£777 million [$1,049 million] for non-elective admissions), according to Low et al, (2015).11 Cost-effective interventions to reduce the burden of hospitalization on patients, caregivers, and healthcare systems should be urgently sought.11
Today, assessment of patients with PD largely depends on motor evaluations performed by physicians during patient visits. However, this approach limits the ability to monitor continuously and accurately track symptom fluctuations or progression of PD in the intervals between scheduled appointments. Ultimately, this makes it more challenging to develop a comprehensive understanding of the patient’s condition over time.
Mobile applications have revolutionized the experience for patients with PD, offering valuable disease-related information, symptom tracking, medication reminders, and tools that assist with daily activities.12 Mobile applications could be useful for managing PD. For patients, mobile applications can be used to monitor symptoms at home and as medication reminders.13 For healthcare professionals, mobile applications enable objective assessment of key parameters and facilitate treatment plans. Mobile applications can enhance the collaboration between healthcare professionals and patients toward more person-centered care and outcome measurements.14
To our knowledge, published systematic reviews have focused on the use of technology and mobile applications in PD for a patient’s self-care and symptom monitoring at home,13 but few have addressed their potential role as digital biomarkers in PD. Although many mobile applications have been developed for PD, their effectiveness in clinical outcomes is inconclusive. Therefore, the aim of this study was to systematically review the effectiveness of mobile applications for PD in diagnosis and self-management. In addition, we conducted meta-analyses to quantitatively determine whether mobile applications result in significant improvements in the clinical outcomes of PD.
Materials and Methods
This systematic review was conducted in accordance with the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 (PRISMA) statement15 and was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under protocol number CRD420251156480.
Search Strategy and Selection Criteria
A search was conducted in PubMed, Ovid Medline, Scopus, ScienceDirect, Cochrane Library, and EMBASE using the search query on November 10, 2024. The terms ‘Parkinson’ AND ‘mobile application’ were used as the keywords for the concept and MeSH to search within the title, abstract, and keywords of the articles.
Inclusion and Exclusion Criteria
All articles published between 2011 and 2024 were included. This start date was chosen because the first reference related to the management of PD by using a mobile application was published in 2011.16 All patients older than 18 years and diagnosed with idiopathic PD were selected.
The inclusion criteria for articles in this systematic review were (1) patients diagnosed with idiopathic PD; (2) mobile applications installed in smartphones with/without supplementary tools such as accelerometers, gyroscopes, smartwatches, and others for medical professionals, patients, and/or caregivers in optimizing patient outcome in PD; (3) articles in English; and (4) original articles published in electronic databases. Abstracts/conference proceedings relevant to mobile applications in PD management were screened. Studies presented in systematic literature reviews, abstracts, case reports, letters to editors, preprint papers, protocols, and surveys were excluded.
Study Selection
The process of the article selection was initiated by screening titles and abstracts identified through the search strategy’s keywords. The articles were downloaded into EndNote reference management software. Duplicate articles were removed with EndNote and manually. Two authors independently reviewed the full texts of the retrieved articles using predefined inclusion and exclusion criteria. The final articles for inclusion were selected after reading the full text of the eligible articles and the references. Any discrepancies between authors were resolved through discussion or by consulting the third author.
Quality Assessment
The quality of included studies was assessed by using the Cochrane risk-of-bias tool. A revised tool for assessing risk of bias in randomized trials (RoB 2) was used for randomized controlled studies.17 Cochrane Risk of Bias in Nonrandomized Studies of Interventions (ROBINS-I) was used to assess risk of bias in nonrandomized studies of interventions.18 The authors independently assessed the quality of the studies, and any discrepancies were resolved by consensus.
Results
Search Results
A total of 2,174 articles were identified through electronic database searches, comprising PubMed (217), Embase (1,042), OVID Medline (211), Cochrane Library (98), ScienceDirect (414), and Scopus (192). After removing duplicates, 1,199 articles remained for screening. Based on title and abstract screening, 975 articles were excluded for not meeting the inclusion criteria. The full texts of 222 articles were retrieved and assessed for eligibility. After reviewing the full text of the articles, the authors reached a consensus to include 19 studies in the final review (Figure 1).
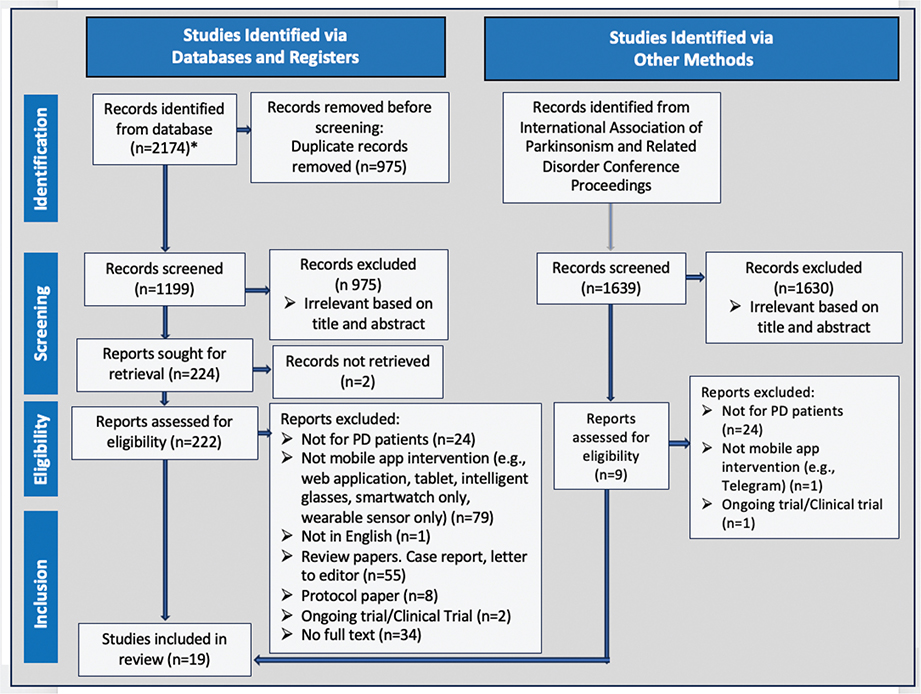
Fig. 1. Flow diagram for selection process. * Cochrane Library = 98, EMBASE = 1042, OVID Medline = 211, PubMed = 217, ScienceDirect = 414, Scopus = 192.
General Characteristics of Included Studies
Appendix A lists the characteristics and outcomes of the included studies. Most studies were cross-sectional, with only four being randomized controlled trials. The duration of monitoring varied widely, ranging from several minutes in clinic settings to up to 1 year in home-based environments.
The use of mobile applications is summarized in Figure 2. In the majority of studies, patients used mobile applications at home. Among these, 10 studies focused on self-monitoring of PD symptoms,19–28 and one examined ambulatory data during the ‘ON’ phase (after medication intake) and ‘OFF’ phase (following medication withdrawal).29 One study evaluated changes in quality of life,30 one assessed the impact of smartphone-based education,31 two investigated smartphone-based exercise programs,32,33 and one explored smartphone-assisted rehabilitation.34 In addition, three studies used mobile applications for the finger tapping test performance and walking test for clinic patients.29,35,36
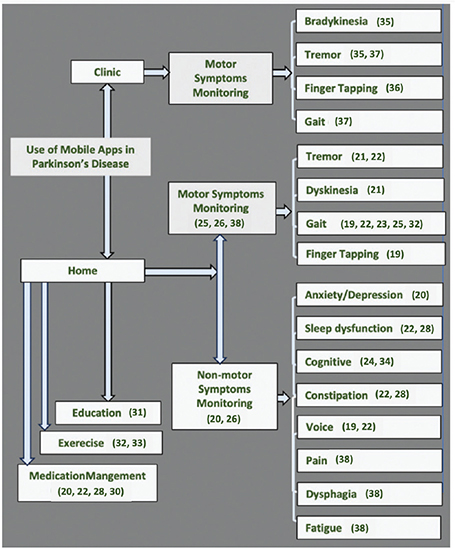
Fig. 2. Use of mobile applications in Parkinson’s disease.
Study Quality Assessment
The risk-of-bias assessments across five domains for four randomized studies are based on RoB 2. Two studies had an overall low risk of bias,26,34 while two raised concerns, primarily related to exclusion from the analysis due to missing outcome data20 and the potential for selective outcome reporting, particularly when only feasibility or preliminary efficacy outcomes were highlighted in the pilot study27 (Table 1).
Table 1. The risk-of-bias assessments across five domains for four randomized studies
Table 2 summarizes the risk of bias assessment across seven ROBINS-I domains for 15 nonrandomized studies. None of the studies achieved low overall risk of bias. Ten studies were rated as serious risk,19,21,23–25,28-30,32,37 mainly due to unmeasured, confounding, and incomplete outcome data, while five were rated moderate.22,31,33,35,36
Table 2. The risk of a biased assessment across seven ROBINS-I domains for 15 nonrandomized studies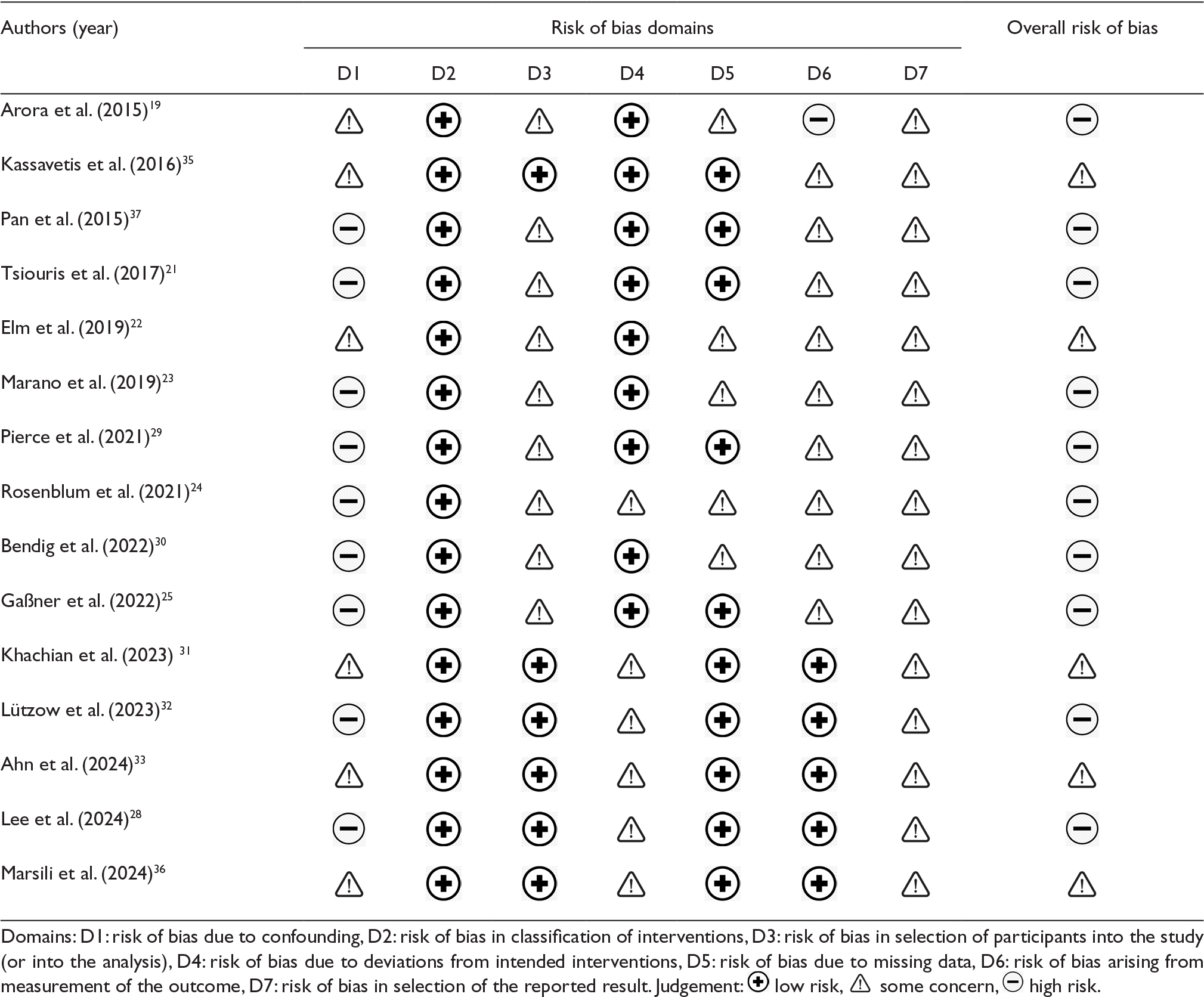
The risk of bias is common in observational or quasi-experimental studies because most patients are recruited based on convenience or voluntary methods, which can lead to selection bias. In digital health research, blinding of subjects is difficult, and confounding factors such as digital literacy might also affect the research outcome. High dropout rates in some digital health research are due to technical problems or incomplete data in the mobile application. For mobile applications that involve self-reporting by patients, the outcome depends on the individual, which might lead to bias in the measurement of outcomes.
Tools
Most studies used smartphone applications without assistive tools to monitor motor symptoms (finger tapping, 3-m timed-up-and-go test, and static balance test), nonmotor symptoms (cognitive impairment, pain, constipation, dysphagia, sleep disturbances, anxiety, depression, and fatigue), medication management, smartphone-based exercise programs, and self-management education.
Some studies used smartphone applications with assistive tools such as wrist and insole sensors21,25 to monitor gait, tremor, dyskinesia, and bradykinesia; foot-mounted inertial measurement units27 to monitor gait and balance; smartwatches22,26 to monitor motor and nonmotor symptoms, medication compliance, and symptom severity; multimodal telemedical intervention that combines video visits, a smartphone app, a camera system, and wearable sensors30 to monitor effects on medication and supportive therapy; and telerehabilitation plus a virtual reality program34 using smartphone applications to monitor cognitive and social function.
Sensitivity and Accuracy of Mobile Applications in PD
By using a smartphone application, Arora et al. found that the mean sensitivity was 96.2% (standard deviation [SD] 2) and the mean specificity was 96.9% (SD 1.9%) in differentiating patients with PD versus healthy subjects.19 Pan et al.37 demonstrated that for hand resting tremor detection by using a mobile application, the sensitivity was 77% and the accuracy was 82%. Pierce et al. showed that an accuracy of up to 92.5% in gait classification was obtained from smartphone data.29
Meta-Analysis
Among the four randomized studies included in the systematic review, two studies used motor symptoms (UPDRS-III) and two studies used nonmotor symptoms as assessment tools. The results of the random-effect model using RevMan are presented in Table 3. Meta-analysis of four randomized controlled trials (two targeting motor and two targeting nonmotor symptoms) showed no significant difference between mobile application interventions and control groups (overall standard mean difference = –1.12, 95% confidence interval [CI]: –5.53 to 3.29, p = 0.62). Subgroup analyses revealed no significant effect for either motor (standard mean difference = 0.02, 95% CI: –0.41 to 0.46, p = 0.91) or nonmotor symptoms (standard mean difference = –777.73, 95% CI: –2306.27 to 750.80, p = 0.32).
Table 3. The effects of Parkinson’s disease mobile application on motor symptoms (UPDRS-III) and nonmotor symptoms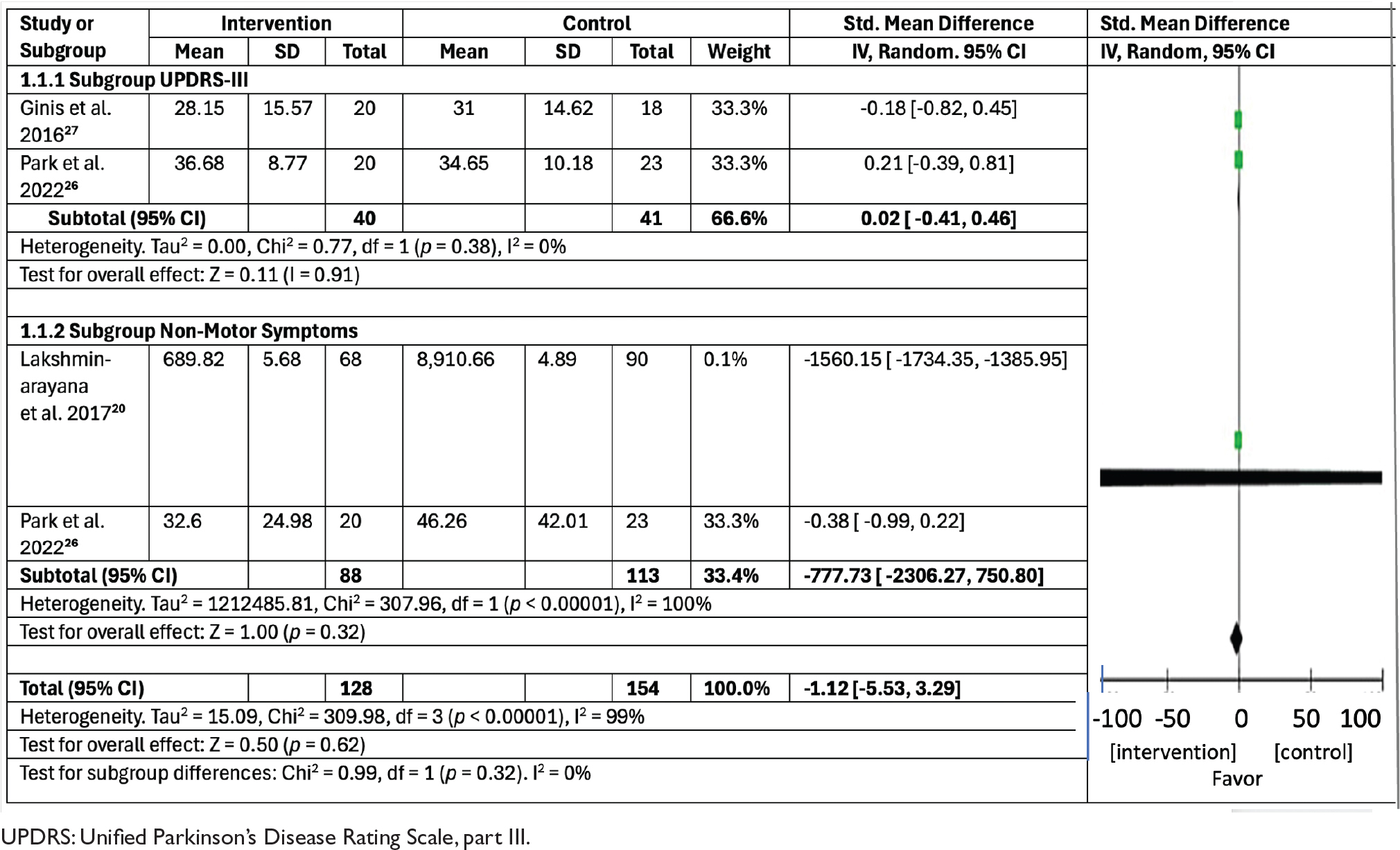
Discussion
To our knowledge, this study is the first systematic review with meta-analysis to assess the impact of mobile applications in PD management.
Morgan et al.13 was the first systematic review to evaluate the use of wearable and nonwearable technologies that passively monitor the activities of patients in the home or home-like environments. However, this systematic review did not clarify whether mobile applications for PD resulted in any improvement of clinical outcomes. Another systematic review by Lee et al.38 found that evidence on the use of mobile applications in patients with PD is still inadequate to establish whether there is any improvement in symptoms, activity levels, medication adherence, or quality of life.
We identified 19 studies on the use of mobile applications in patients with PD. Most applications were designed for symptom monitoring, while others supported diagnostic purposes, exercise, patient education, and medication management. Of the 16 studies that included follow-up of patients using mobile applications, 11 reported significant improvements in clinical outcomes, including physical activity, nonmotor symptoms, self-efficacy, medication adherence, and cognitive function.20,23,25–28,31–34,36 Evidence from the included studies suggests that mobile applications hold potential as supportive tools in the management of PD, particularly in enhancing patient engagement and facilitating remote monitoring. However, most of the studies were nonrandomized and involved small sample sizes and short durations. In addition, the majority of nonrandomized studies were rated as having a moderate to severe risk of bias according to the ROBINS-I V2 assessment tool, which limits the reliability and generalizability of the findings.
Several studies reported the use of mobile applications in monitoring motor symptoms of patients with PD, including bradykinesia,35 tremor,35,37 gait,19,21,29,37 and the finger tapping test.36 Across the included studies,19,21,29,37 the sensitivity and accuracy of mobile applications in measuring gait among patients with PD varied substantially, ranging from 69 to 96%. Mobile applications might serve as a useful screening aid and support diagnostic evaluation, but none have been validated and used as definitive diagnostic instruments.
Mobile applications have been used for self-monitoring of motor and nonmotor symptoms by patients with PD at home. Adherence of the patient and caregiver to mobile applications is crucial. In the included studies, adherence to mobile applications among patients with PD demonstrated considerable variability, with rates ranging from 33 to 100%.19,20,22,25,27,28,30,32–34 These results should be interpreted with caution because most of the studies were short-term and therefore had insufficient data to make any conclusion about patients’ adherence to mobile applications in the long term.
Patients with PD who have motor impairment, such as tremor, might have difficulty using mobile applications, especially during the OFF phase of anti-Parkinson’s medication. Most of the patients with PD are elderly and might have limited digital literacy, which will also affect their adherence to mobile applications. Besides, the complexity of the task required by the patient can reduce the usability and engagement of the mobile application. For example, patients were asked to monitor the motor symptoms every 30 min over 3 days in the electronic diary30 or rate the symptom severity (for up to eight types of symptoms) three times per day,22 which led to low adherence by patients to the mobile application. In addition, if clinicians are not engaged at the early stages of mobile application development, it might result in a gap in mobile application measures and clinical assessments, which might limit the tool’s credibility and acceptance in practice.22
Although the mobile applications in the 19 studies were independently developed, there was a high degree of similarity between their functions. Most applications focused on motor symptoms such as tremor (n = 4), finger tapping (n = 2), and gait disturbances (n = 6). In addition, others covered nonmotor symptoms such as sleep disturbances (n = 2), cognitive impairment (n = 2), constipation (n = 2), and voice changes (n = 2) (Figure 2).
Apart from monitoring motor or nonmotor symptoms by using mobile applications, some features supported medication adherence, patient education, and exercise programs. A major limitation is that none bring together all relevant functions into a single platform. This means patients might need to use more than one application at the same time, which will reduce patients’ adherence to mobile applications, especially those with pre-existing motor and cognitive impairments. There is potential for collaboration on mobile application development, but coordination between different research teams and regulatory bodies remains challenging. Some studies used clinical measurement tools such as the unified Parkinson’s disease rating scale (UPDRS) for motor assessment of patients with PD, but the tools in mobile applications need to be validated before being used in routine clinical practice.
Our systematic review included a meta-analysis of randomized controlled trials to quantitatively determine the impact of mobile applications on clinical outcomes of PD. Meta-analytic results showed that current evidence was not sufficient to show any positive benefit of mobile applications in improving clinical outcomes among patients with PD. However, larger randomized controlled trials are needed to acquire results that will be more robust and reliable.
Future Directions
A roadmap is crucial to enforce patient-centered digital outcome measures through mobile health technologies. Such a roadmap has been proposed by the Technical Working Group of the Movement Disorders Association, which comprises an online survey to determine patient-relevant endpoints, criteria on combination devices, examples of open-source integration platforms, and guidelines on assessments of regulatory readiness concerning new devices.39 Research teams must collaborate to conduct higher-quality and larger-scale randomized studies. Given similarities in functional features between mobile applications for PD, this collaboration will greatly assist research teams in creating better quality mobile applications and reducing redundancy. Besides, the involvement of doctors, patients, and caregivers with research teams at the initial stage of mobile application development is important. This ensures that mobile applications are addressing end-user needs and are sustainable for integration into the daily life of the patient in the long term.
Mobile applications can be considered as potential digital biomarkers for PD only after validation with reference tools (e.g. gyroscopes/accelerometers/inertial sensors) and clinical scales (e.g. UPRDS). Besides, mobile applications should be validated for accuracy (laboratory validity), reliability (test-retest within and across sensors), sensitivity, and minimal clinically significant difference for relevant endpoints when evaluated with patient condition.39 Mobile application development should also comply with emerging regulatory requirements on digital health technologies. Health data are sensitive personal data; therefore, security, transparency, and accountability are three key aspects of building systems for the protection of health data. National guidelines on patient rights should also be considered to safeguard patients and effectively prevent unauthorized access or misuse and loss of data.40
Future research on PD mobile applications should explicitly consider medication status, such as through standardized measurement of drug action periods (ON or OFF) or stratified analysis, for valid interpretation of intervention effects. This systematic review contains several studies analyzing the effects of smartphone-based training interventions on motor function in patients with PD.27,32,33 However, a major limitation is that most studies do not clearly specify whether the assessment of motor outcomes was conducted during the ON or OFF phase of anti-Parkinson’s medication. Motor function fluctuates significantly with dopaminergic therapy cycles,41 and not controlling medication status might lead to potential confounding whereby improvements in reported motor function could be due to differences in medication timing (ON or OFF phase) rather than the actual effect due to the intervention.
The sustainability of PD mobile applications needs to be taken into consideration because it involves software updates, cybersecurity measures, user support, and adaptation to clinical practices. All these requirements will incur additional costs that might affect the expansion of the mobile application, especially in healthcare systems with limited resources. Therefore, economic analysis can be carried out to assess whether the mobile application causes a positive net benefit to the healthcare system.
To date, completed cost-utility evaluations (such as quality-adjusted life year [QALY]-based specifically for mobile applications in PD) remain scarce, with most mobile application studies reporting feasibility or clinical endpoints rather than economics. Muñoz et al. found that early detection of PD using a sensor-based intervention has an estimated cost-effectiveness of $31,305/QALY, which is lower than the commonly accepted threshold used in the U.S.,42 but this study did not involve a mobile application. Another study investigated the costs and results of telerehabilitation in neurological (particularly in stroke patients) and cardiological diseases, but not in PD.43
In the future, the development of artificial intelligence (AI)-driven mobile applications can be explored to facilitate early detection, symptom monitoring, and treatment of PD. A recent systematic review highlighted the use of an AI-based approach in PD diagnosis, with the accuracy rates between 78 and 96%.44 Besides, AI has been used to assess PD severity in a home setting and enable remote evaluation of patients by a doctor.45
Strengths and Limitations
This is the first systematic review to unify and consolidate existing evidence on mobile app-based diagnostics and self-management interventions for PD in a single comprehensive synthesis. Slightly different from previous reviews that emphasized either symptom monitoring or patient self-management, this article discussed the emerging role of mobile apps as digital biomarkers, expanding the clinically relevant dimensions of telehealth in PD.
The review was conducted using a rigorous and transparent methodology, a structured literature search, predefined inclusion criteria, and a standardized risk of bias assessment according to PRISMA guidelines. Both cross-sectional studies and randomized controlled trials reflected real-world, large-scale assessments of clinical effectiveness. The use of meta-analysis allowed for quantitative estimation of the number of patients achieving specific clinical outcomes, thus providing an objective evidence base that goes beyond narrative synthesis.
Several limitations should be noted. A limited number of randomized controlled trials were conducted, and significant heterogeneity was observed across studies in terms of study design, outcome measures, follow-up duration, and mobile app functionality, which may have impacted the robustness of the pooled estimates. Many of the included studies were cross-sectional, limiting causal inferences regarding clinical effectiveness.
Variation in the validation status of mobile apps and outcome measures, particularly those related to digital biomarkers, might have introduced measurement error. This review might also lack relevant studies due to publication bias and language limitations. Because this systematic review only included the studies with PD mobile applications, interventions that involve other digital health and telemedicine were excluded. Furthermore, studies published before 2011 were excluded, which might result in missing relevant articles from this period.
Conclusion
Due to the limited number and heterogeneity of existing studies, the evidence regarding the effectiveness of mobile applications in the treatment of PD remains inconclusive. Further research is needed to strengthen the evidence and assess the clinical benefits of mobile applications in the management of PD.
Contributions
Mei Wah Lee contributed conceptualization, data curation, formal analysis, methodology, visualization, and writing the original draft. Umar Idris Ibrahim contributed conceptualization, data curation, formal analysis, methodology, visualization, writing, review, and editing. Pei Lin Lua contributed conceptualization, data curation, formal analysis, methodology, visualization, supervision, and writing—review and editing. All authors approved the final article.
Data Availability Statement (DAS), Data Sharing, Reproducibility, and Data Repositories
See the Appendix.
Application of AI-Generated Text or Related Technology
This article is not AI generated text.
References
- Kalia LV, Lang AE. Parkinson’s disease. Lancet. 2015;386:896–912. https://doi.org/10.1016/S0140-6736(14)61393-3
- Obeso JA, Stamelou M, Goetz CG, Poewe W, Lang AE, Weintraub D, et al. Past, present, and future of Parkinson’s disease: a special essay on the 200th Anniversary of the Shaking Palsy. Mov Disord. 2017;32:1264–310. https://doi.org/10.1002/mds.27115
- Dorsey ER, Constantinescu R, Thompson JP, Biglan KM, Holloway RG, Kieburtz K, et al. Projected number of people with Parkinson’s disease in the most populous nations, 2005 through 2030. Neurology. 2007;68(5). https://doi.org/10.1212/01.wnl.0000247740.47667.03
- Schapira AHV, Chaudhuri KR, Jenner P. Non-motor features of Parkinson’s disease. Nat Rev Neurosci. 2017;18:435–50. https://doi.org/10.1038/nrn.2017.62
- Zhao N, Yang Y, Zhang L, Zhang Q, Balbuena L, Ungvari GS, et al. Quality of life in Parkinson’s disease: a systematic review and meta-analysis of comparative studies. CNS Neurosci Ther. 2021;27:270–9. https://doi.org/10.1111/cns.13549
- Al-Khammash N, Al-Jabri N, Albishi A, Al-Onazi A, Aseeri S, Alotaibi F, et al. Quality of life in patients with Parkinson’s disease: a cross-sectional study. Cureus. 2023 Jan 20;15(1):e33989. https://doi.org/10.7759/cureus.33989. PMID: 36824559; PMCID: PMC9941031.
- Hoseinipalangi Z, Kan FP, Hosseinifard H, Doustmehraban M, Masoumi M, Rafiei S, et al. Systematic review and meta-analysis of the quality-of-life of patients with parkinson’s disease. East Mediterr Health J. 2023;29:63–70. https://doi.org/10.26719/emhj.23.013
- Yi ZM, Willis S, Zhang Y, Liu N, Tang QY, Zhai SD. Impact of a collaborative pharmaceutical care service for patients with Parkinson’s disease. Front Pharmacol. 2022 Jan 3;12:793361. https://doi.org/10.3389/fphar.2021.793361. PMID: 35046815; PMCID: PMC8762333.
- Jasinska-Myga B, Heckman MG, Wider C, Putzke JD, Wszolek ZK, Uitti RJ. Loss of ability to work and ability to live independently in Parkinson’s disease. Parkinsonism Relat Disord. 2012;18(2):130–5. https://doi.org/10.1016/j.parkreldis.2011.08.022
- Gerlach OHH, Winogrodzka A, Weber WEJ. Clinical problems in the hospitalized Parkinson’s disease patient: systematic review. Mov Disord. 2011;26:197–208. https://doi.org/10.1002/mds.23449
- Low V, Ben-Shlomo Y, Coward E, Fletcher S, Walker R, Clarke CE. Measuring the burden and mortality of hospitalisation in Parkinson’s disease: a cross-sectional analysis of the English Hospital Episodes Statistics database 2009–2013. Parkinsonism Relat Disord. 2015;21(5):449–54. https://doi.org/10.1016/j.parkreldis.2015.01.017
- Sjödahl Hammarlund C, Westergren A, Åström I, Edberg AK, Hagell P. The impact of living with Parkinson’s disease: balancing within a web of needs and demands. Parkinsons Dis. 2018;2018:4598651. https://doi.org/10.1155/2018/4598651
- Morgan C, Rolinski M, McNaney R, Jones B, Rochester L, Maetzler W, et al. Systematic review looking at the use of technology to measure free-living symptom and activity outcomes in parkinson’s disease in the home or a home-like environment. J Parkinsons Dis. 2020;10:429–54. https://doi.org/10.3233/JPD-191781
- Luis-Martínez R, Monje MHG, Antonini A, Sánchez-Ferro Á, Mestre TA. Technology-enabled care: integrating multidisciplinary care in Parkinson’s disease through digital technology. Front. Neurol. 2020; 11:575975. https://doi.org/10.3389/fneur.2020.575975
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. https://doi.org/10.1136/bmj.n71
- Linares-del Rey M, Vela-Desojo L, Cano-de la Cuerda R. Mobile phone applications in Parkinson’s disease: a systematic review. Neurologia. 2019;34:38–54. https://doi.org/10.1016/j.nrl.2017.03.006
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366. https://doi.org/10.1136/bmj.l4898
- Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016; 355:i4919. https://doi.org/10.1136/bmj.i4919
- Arora S, Venkataraman V, Zhan A, Donohue S, Biglan KM, Dorsey ER, et al. Detecting and monitoring the symptoms of Parkinson’s disease using smartphones: a pilot study. Parkinsonism Relat Disord. 2015;21(6):650–3. https://doi.org/10.1016/j.parkreldis.2015.02.026
- Lakshminarayana R, Wang D, Burn D, Chaudhuri KR, Galtrey C, Guzman NV, et al. Using a smartphone-based self-management platform to support medication adherence and clinical consultation in Parkinson’s disease. NPJ Parkinsons Dis. 2017;3(1). https://doi.org/10.1038/s41531-017-0034-0
- Tsiouris KM, Gatsios D, Rigas G, Miljkovic D, Seljak BK, Bohanec M, et al. PD-Manager: an mHealth platform for Parkinson’s disease patient management. Healthc Technol Lett. 2017;4(3):102–8. https://doi.org/10.1049/htl.2017.0007
- Elm JJ, Daeschler M, Bataille L, Schneider R, Amara A, Espay AJ, et al. Feasibility and utility of a clinician dashboard from wearable and mobile application Parkinson’s disease data. NPJ Digit Med. 2019 Sep 25;2:95. https://doi.org/10.1038/s41746-019-0169-y. PMID: 31583283; PMCID: PMC6761168.
- Marano M, Motolese F, Rossi M, Magliozzi A, Yekutieli Z, Di Lazzaro V. Remote smartphone gait monitoring and fall prediction in Parkinson’s disease during the COVID-19 lockdown. Neurol Sci. 2021;42(8):3089–92. https://doi.org/10.1007/s10072-021-05351-7
- Rosenblum S, Richardson A, Meyer S, Nevo T, Sinai M, Hassin-Baer S. Dailycog: a real-world functional cognitive mobile application for evaluating mild cognitive impairment (mci) in Parkinson’s disease. Sensors. 2021;21(5):1–14. https://doi.org/10.3390/s21051788
- Gaßner H, Friedrich J, Masuch A, Jukic J, Stallforth S, Regensburger M, et al. The effects of an individualized smartphone-based exercise program on self-defined motor tasks in Parkinson’s disease: pilot interventional study. JMIR Rehabil Assist Technol. 2022;9(4). https://doi.org/10.2196/38994
- Park Y, Kim SR, So HY, Jo S, Lee SH, Hwang Y, et al. Effect of mobile health intervention for self-management on self-efficacy, motor and non-motor symptoms, self-management, and quality of life in people with Parkinson’s disease: randomized controlled trial. Geriatr Nurs (Minneap). 2022;46:90–7.
- Ginis P, Nieuwboer A, Dorfman M, Ferrari A, Gazit E, Canning CG, et al. Feasibility and effects of home-based smartphone-delivered automated feedback training for gait in people with Parkinson’s disease: a pilot randomized controlled trial. Parkinsonism Relat Disord. 2016;22:28–34. https://doi.org/10.1016/j.parkreldis.2015.11.004
- Lee JH, Suh Y, Kim E, Yoo S, Kim Y. A mobile app for comprehensive symptom management in people with Parkinson’s disease: a pilot usability study. Comput Inform Nurs. 2024;42(4):289–97.
- Pierce A, Ignasiak NK, Eiteman-Pang WK, Rakovski C, Berardi V. Mobile phone sensors can discern medication-related gait quality changes in Parkinson’s patients in the home environment. Computer Methods and Programs in Biomedicine Update. 2021;1. 100028. https://doi.org/10.1016/j.cmpbup.2021.100028
- Bendig J, Wolf AS, Mark T, Frank A, Mathiebe J, Scheibe M, et al. Feasibility of a multimodal telemedical intervention for patients with Parkinson’s disease—a pilot study. J Clin Med. 2022;11(4). https://doi.org/10.3390/jcm11041074
- Khachian A, Zeighami K, Khoubbin Khoshnazar TA, Haghani H. The effectiveness of self-management education based on the mobile application on the quality of life of people with parkinson’s disease. Middle East J Rehabil Health Stud. 2023;10(2). https://doi.org/10.5812/mejrh-131057
- Lützow L, Teckenburg I, Koch V, Marxreiter F, Jukic J, Stallforth S, et al. The effects of an individualized smartphone-based exercise program on self-defined motor tasks in Parkinson’s disease: a long-term feasibility study. J Patient Rep Outcomes. 2023;7(1). https://doi.org/10.1186/s41687-023-00631-6
- Ahn JH, Shin D, Lee D, Kim HY, Youn J, Cho JW. Increasing exercise with a mobile app in people with Parkinson’s disease: a pilot study. Brain Impair. 2024;25(2).
- Maggio MG, Luca A, Cicero CE, Calabrò RS, Drago F, Zappia M, et al. Effectiveness of telerehabilitation plus virtual reality (Tele-RV) in cognitive e social functioning: a randomized clinical study on Parkinson’s disease. Parkinsonism Relat Disord. 2024;119. https://doi.org/10.1016/j.parkreldis.2023.105970
- Kassavetis P, Saifee TA, Roussos G, Drougkas L, Kojovic M, Rothwell JC, et al. Developing a tool for remote digital assessment of Parkinson’s disease. Mov Disord Clin Pract. 2016;3(1):59–64.
- Marsili L, Abanto J, Mahajan A, Duque KR, Chinchihualpa Paredes NO, Deraz HA, et al. Dysrhythmia as a prominent feature of Parkinson’s disease: an app-based tapping test. J Neurol Sci. 2024;463. https://doi.org/10.1016/j.jns.2024.123144
- Pan D, Dhall R, Lieberman A, Petitti DB. A mobile cloud-based Parkinson’s disease assessment system for home-based monitoring. JMIR Mhealth Uhealth. 2015;3(1):e29. https://doi.org/10.2196/mhealth.3956
- Lee JH, Yeom I, Chung ML, Kim Y, Yoo S, Kim E. Use of mobile apps for self-care in people with Parkinson’s disease: systematic review. JMIR mHealth uHealth. 2022;10. https://doi.org/10.2196/33944
- Espay AJ, Hausdorff JM, Sánchez-Ferro Á, Klucken J, Merola A, Bonato P, et al. A roadmap for implementation of patient-centered digital outcome measures in Parkinson’s disease obtained using mobile health technologies. Mov Disord. 2019;34:657–63. https://doi.org/10.1002/mds.27671
- Global strategy on digital health 2020–2025. World Health Organization; 2021. Licence: CC BY-NC-SA 3.0 IGO.
- Yin W, Gao H, Liang B, Liu R, Liu Y, Shen C, et al. Quantitative analysis of gait parameters in Parkinson’s disease and the clinical significance. Front Neurol. 2025;16. https://doi.org/10.3389/fneur.2025.1527020
- Muñoz DA, Kilinc MS, Nembhard HB, Tucker C, Huang X. Evaluating the cost-effectiveness of an early detection of Parkinson’s disease through innovative technology. Eng Econ. 2017;62(2):180–96. https://doi.org/10.1080/0013791X.2017.1294718
- Del Pino R, Díez-Cirarda M, Ustarroz-Aguirre I, Gonzalez-Larragan S, Caprino M, Busnatu S, et al. Costs and effects of telerehabilitation in neurological and cardiological diseases: a systematic review. Front Med. 2022;9. https://doi.org/10.3389/fmed.2022.832229
- Twala B. AI-driven precision diagnosis and treatment in Parkinson’s disease: a comprehensive review and experimental analysis. Front Aging Neurosci. 2025;17. https://doi.org/10.3389/fnagi.2025.1638340
- Islam MS, Rahman W, Abdelkader A, Lee S, Yang PT, Purks JL, et al. Using AI to measure Parkinson’s disease severity at home. NPJ Digit Med. 2023;6(1). https://doi.org/10.1038/s41746-023-00905-9
Appendix A: A summary of the studies included in the review
| Author, Year, and Country | Study design | Sample size | Tool | Intervention duration | Location | Assessment | Results |
| Arora et al. (2015) / United States19 | Cross-sectional study | 10 PD patients and 10 controls | Smartphone | Five tasks four times a day for a month | Home | Voice, posture, gait, finger tapping, and response time | In discriminating participants with PD from controls, the mean sensitivity was 96.2% (SD 2%) and the mean specificity was 96.9% (SD 1.9%). |
| Kassavetis et al. (2016) / London, UK35 | Cross-sectional study | 14 PD patients | Smartphone with accelerometer | A single experimental session lasted approximately 30 min. | Clinic | Tremor, bradykinesia | Significant correlation between five subscores of MDS-UPDRS (rest tremor, postural tremor, pronation-supination, leg agility, and finger tapping) and eight parameters of the data collected with the smartphone. |
| Pan et al. (2015) / United States37 | Cross-sectional study | 40 PD patients | Smartphone with accelerometer | A single motor performance test session | Clinic | Hand resting tremor, walking, and turning | For hand resting tremor detection, the sensitivity was 0.77, and the accuracy was 0.82. For gait difficulty detection, the sensitivity was 0.89, and the accuracy was 0.81. |
| Lakshminarayana et al. (2017) / England, Scotland20 | RCT | 94 PD patients and 107 controls | Smartphone | 16 weeks | Home | Medication adherence, QoL, nonmotor symptoms, anxiety, depression, beliefs about medication | Parkinson’s tracker app significantly improved adherence, compared with treatment as usual (mean difference: 0.39, 95% CI: 0.04–0.74; p = 0.0304), patients’ perception of quality of consultation (0.15, 95% CI: 0.03 to 0.27; p = 0.0110). |
| Tsiouris et al. (2017) / Europe21 | Cross-sectional study | 20 PD patients | Smartphone, commercial wrist and insole sensors | Eight sessions in which patients performed a set of everyday activities following the protocol | Home | Motor symptoms (gait, freezing of gait events, tremor, dyskinesia, and bradykinesia) | Gait: 69% was correctly estimated in accordance to the clinician’s MDS-UPDRS gait annotation. Tremor: detected with 94% accuracy, while amplitude estimation had an 88% accuracy across all classes Dyskinesia: detection accuracy was between 88 and 98% |
| Elm et al. (2019) / United States22 | Cross-sectional study | 39 PD patients | Clinician dashboard, smartphone, smartwatch | 6 months Each participant was asked to report their symptoms (for up to eight types of symptoms) at least three times per day in the app. |
Home | Constipation, balance, rigidity, bradykinesia, voice, dyskinesia, tremor, gait, sleep dysfunction, medication compliance, symptom severity | Reporting of symptoms and medication intake using the app was lower than expected, 44 and 60%, respectively. |
| Marano et al. (2021) / Italy23 | Cross-sectional study | 33 PD patients | Smartphone | 4 weeks | Home | 3-min timed-up-and-go test static balance test | The stand-up time and the mediolateral sway, acquired through the application, differed between ‘fallers’ and ‘nonfallers’ and related to the occurrence of new falls (OR 1.7 and 1.6, respectively, p < 0.05), together with previous falling (OR 7.5, p < 0.01). |
| Pierce et al. (2021) / USA29 | Cross-sectional study | 20 PD patients | Smartphone | Two observation sessions (i.e. OFF vs ON) | Home | Ambulatory data after medication abstention (OFF) and approximately 30 min after intake of their medication (ON) | Up to 92.5% gait classification accuracy was achieved using raw smartphone data. |
| Rosenblum et al. (2021) / Israel24 | Cross-sectional study | 36 PD patients | Smartphone | The two evaluation tasks were carried out with an interval of 3–7 months between each task | Home | Mild cognitive impairment | Did not obtain statistical significance for the differences between users with high and low MoCA scores |
| Bendig et al. (2022) / Germany30 | Cross-sectional study | 11 PD patients | Multimodal telemedical intervention that combines video visits, a smartphone app, a camera system, and wearable sensors | 12 weeks | Home | QoL, effects on medication and supportive therapy | No significant difference in the total PDQ-39 score between the first and the second measurement (p = 0.9972, paired t-test). |
| Gaßner et al. (2022) / Germany25 | Cross-sectional study | 15 PD patients | Smartphone, wearable sensors that were attached to the outer rear side of each shoe | 4 weeks | Home | System usability scale, QoL, patient-defined motor symptoms, UPDRS-III, timed up and go test, 2-min walking test, and sensor-based gait analysis. | Patient-defined motor tasks of daily living significantly improved by 40% on average in 87% (13/15) of the patients. |
| Park et al. (2022) / South Korea26 | RCT | 20 PD patients and 23 controls | Mobile applications, smartwatches, smartphone-based short text messages and information, and telephone counselling | 16 weeks | Home | Self-efficacy, motor symptom, nonmotor symptom, self-management, QoL | Self-efficacy and nonmotor symptom scores in the intervention group significantly improved compared with those in the control group. |
| Khachian et al. (2023) / Iran31 | Cross-sectional study | 77 PD patients | Smartphone | 12 weeks | Home | Effect of self-management education based on the mobile application on the QoL | The mean score of QoL was significantly improved |
| Lützow, L et al. (2023) / Germany32 | Cross-sectional study | Total= 33 PD patients (17 in smartphone group and 16 in control group) | Smartphone | 26-week training period | Home | UPDRS‑III, gait parameters, QoL | Did not show any significant changes in UPDRS-III, sensor-based gait parameters, and QoL |
| Ahn et al. (2024) / South Korea33 | Cross-sectional study | 25 PD patients | Smartphone | 2 weeks | Home | Effect of home-based exercise program in mobile apps on physical activity and QoL | Median MET-min/week rose from 1386.0 to 3210.0 (P = 0.009), primarily in moderate activities (P = 0.049) and walking (P = 0.002). Median PDQ-39 scores showed improvement from 17.2 to 8.5 (P = 0.005) after the program. |
| Lee et al. (2023) / Korean38 | Cross-sectional study | 22 PD patients | Smartphone | 6 weeks | Home | Nonmotor symptoms (pain, constipation, dysphagia, sleep disturbances, and fatigue), games to address motor symptoms, and medication management | The successful experience of self-monitoring had improved participants’ self-efficacy (Z = −3.634, P < 0.001) and medication adherence (Z = −3.371, P = 0.001). |
| Maggio et al. (2024) / Italy34 | RCT | 34 PD patients | Telerehabilitation plus virtual reality program using two rehabilitation applications on smartphones | 6 weeks | Home | Cognitive and social function | The intergroup comparison showed that smartphone application groups had significantly greater improvements than active control group in MoCA score. |
| Marsili et al. (2024) / US36 | Cross-sectional study | 295 PD patients and 62 controls | Smartphone | 1 year | Clinic | Tapping test performance: Coefficient of variation, coefficient of quartile variation, time between taps (ms), frequency (taps/s), number of taps, duration (s) | Intertap variability (dysrhythmia) but no intertap intervals or tapping speed are reliably distinctive features of an app-based bradykinesia assessment in PD. |
| Ginis et al. (2016) / Belgium, Israel27 | RCT | Total= 40 PD patients (22 in smartphone group and 18 in control group) | A smartphone application offered positive and corrective feedback on gait with the foot-mounted IMUs | 6 weeks | Home | Gait, balance, endurance, and QoL | The smartphone group improved significantly more on balance at posttest (from 24.8 to 26.1, SD~5) and maintained QoL (SF-36 physical health) at follow-up, whereas the control group deteriorated (from 50.4 to 48.3, SD~16). |
| PD: Parkinson’s disease; SD: standard deviation; IMU: inertial measurement units. PUBMED search strategy. ((“parkinson disease”[MeSH Terms] OR (“parkinson”[All Fields] AND “disease”[All Fields]) OR “parkinson disease”[All Fields] OR “parkinson s”[All Fields] OR “parkinsons”[All Fields] OR “parkinson”[All Fields] OR “parkinsonian disorders”[MeSH Terms] OR (“parkinsonian”[All Fields] AND “disorders”[All Fields]) OR “parkinsonian disorders”[All Fields] OR “parkinsonism”[All Fields] OR “parkinsonisms”[All Fields] OR “parkinsons s”[All Fields]) AND (“mobile applications”[MeSH Terms] OR (“mobile”[All Fields] AND “applications”[All Fields]) OR “mobile applications”[All Fields] OR (“mobile”[All Fields] AND “application”[All Fields]) OR “mobile application”[All Fields])) COCHRANE LIBRARY search strategy. #1 MeSH descriptor: [“Parkinson”] explode all trees #2 “Lewy Body Parkinson’s Disease” OR “Idiopathic Parkinson’s Disease” OR “Parkinson Disease” OR Idiopathic OR “Parkinson’s Disease” OR “Primary Parkinsonism” OR “Parkinson’s Disease, Lewy Body” OR “Paralysis Agitans” OR “Parkinson’s Disease, Idiopathic” OR “Lewy Body Parkinson Disease” OR “Idiopathic Parkinson Disease” OR Parkinsonism OR Primary #3 #1 OR #2 #4 MeSH descriptor: [Mobile Applications] explode all trees #5 “Mobile Application” OR “Apps, Mobile” OR “Mobile Apps” OR “App, Mobile” OR “Application, Mobile” OR “Applications, Mobile” OR “Mobile App” OR “Smartphone Apps” OR “Apps, Smartphone” OR “App, Smartphone” OR “Smartphone App” OR “Portable Electronic Application” OR “Portable Electronic Apps” OR “Electronic App, Portable” OR “App, Portable Electronic” OR “Portable Electronic Applications” OR “Application, Portable Electronic” OR “Electronic Application, Portable” OR “Portable Electronic App” OR “Software Application, Portable” OR “Application, Portable Software” OR “Portable Software Applications” OR “Software App, Portable” OR “Portable Software Apps” OR “App, Portable Software” OR “Portable Software App” OR “Portable Software Application” #6 #4 OR #5 #7 #3 AND #6 EMBASE search strategy. #1 ‘parkinson disease’/exp #2 ‘idiopathic parkinsonism’ OR ‘Lewy bodies of Parkinson disease’ OR ‘Lewy bodies of Parkinson`s disease’ OR ‘Lewy bodies of Parkinsons disease’ OR ‘Lewy body Parkinson disease’ OR ‘Lewy body Parkinson`s disease’ OR ‘Lewy body Parkinsons disease’ OR ‘paralysis agitans’ OR ‘Parkinson dementia complex’ OR ‘Parkinson`s disease’ OR ‘Parkinsons disease’ OR ‘primary parkinsonism’ OR ‘Parkinson disease’ #3 #1 OR #2 #4 ‘mobile application’/exp #5 ‘mobile app’ OR ‘mobile applications’ OR ‘mobile apps’ OR ‘portable software app’ OR ‘portable software application’ OR ‘portable software applications’ OR ‘portable software apps’ OR ‘tablet application’ OR ‘mobile application’ #6 #4 OR #5 #7 #3 AND #6 OVID MEDLINE search strategy. (“parkinson disease”[MeSH Terms] OR (“parkinson”[All Fields] AND “disease”[All Fields]) OR “parkinson disease”[All Fields]) AND (“mobile applications”[MeSH Terms] OR (“mobile”[All Fields] AND “applications”[All Fields]) OR “mobile applications”[All Fields] OR (“mobile”[All Fields] AND “application”[All Fields]) OR “mobile application”[All Fields]) ScienceDirect search strategy. https://www.sciencedirect.com/search?qs=parkinson+and+mobile+application&date=2011-2024&years=2011%2C2012%2C2013%2C2014%2C2015%2C2016%2C2017%2C2018%2C2019%2C2020%2C2021%2C2022%2C2023%2C2024&lastSelectedFacet=publicationTitles&articleTypes=FLA&langs=en&subjectAreas=2800&publicationTitles=280683%2C271171%2C785212%2C271055%2C271071%2C271069%2C786868%2C272509%2C272536%2C271072%2C785210%2C271068%2C272194%2C272508%2C271597%2C271080%2C271029%2C280061%2C272555 Scopus search strategy. TITLE-ABS-KEY ( parkinson AND mobile AND application ) AND PUBYEAR > 2010 AND PUBYEAR < 2025 AND ( LIMIT-TO ( SUBJAREA , “MEDI” ) OR LIMIT-TO ( SUBJAREA , “NEUR” ) OR LIMIT-TO ( SUBJAREA, “HEAL”) OR LIMIT-TO ( SUBJAREA , “PHAR” ) ) AND ( LIMIT-TO ( DOCTYPE , “ar” ) OR LIMIT-TO (DOCTYPE , “re” ) ) AND ( LIMIT-TO ( LANGUAGE , “English” )) |
|||||||
Copyright Ownership: This is an open-access article distributed in accordance with the Creative Commons Attribution Non-Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See http://creativecommons.org/licenses/by-nc/4.0. The authors of this article own the copyright.